Search
Thermo Scientific Chemicals
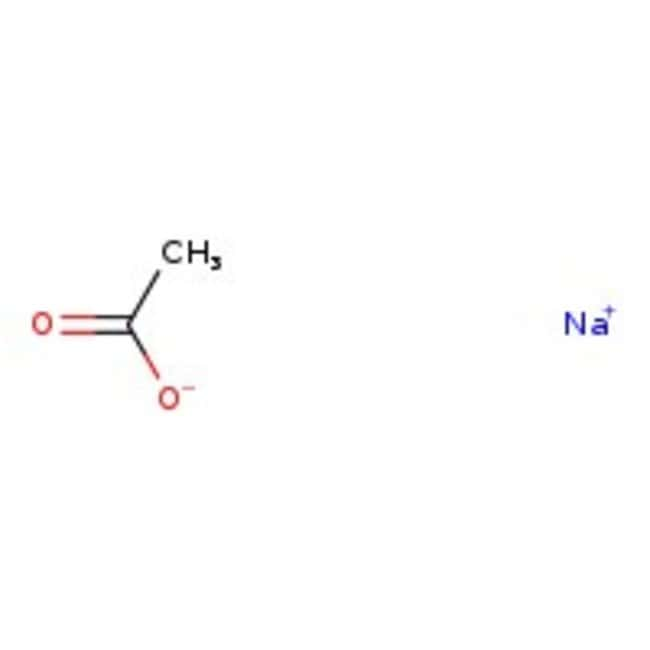
Sodium acetate, anhydrous, 99%
CAS: 127-09-3 | C2H3NaO2 | 82.03 g/mol
化学物質識別子
CAS127-09-3
IUPAC Namesodium acetate
Molecular FormulaC2H3NaO2
InChI KeyVMHLLURERBWHNL-UHFFFAOYSA-M
SMILES[Na+].CC([O-])=O
さらに表示
仕様 スペックシート
スペックシート
FormCrystals or powder or crystalline powder
Identification (FTIR)Conforms
Melting Point>300?C
Assay (Non-aqueous acid-base Titration)≥98.5 to ≤101.5%
Water Content (Karl Fischer Titration)<1.0%
さらに表示
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Suitable for battery materials development.
Applications
Sodium acetate is used as buffering agent with acetic acid. It is used in many areas like cosmetics, pharmaceuticals, agriculture, bronzing and textile industry. It is employed in production of dye materials, as a polymerization catalyst, as a polymer stabilizer, as a plating agent, preparation of gel stains, preservative in food production and flavor enhancer in the nutrient industry. Sodium acetate used in the study of lithography, photography. It is used as a base in several organic reactions in research laboratories and industries.
Solubility
Very soluble in water. Soluble in alcohol.
Notes
Hygroscopic. It is incompatible with strong oxidizing agents, halogens and it is moisture sensitive.
Sodium acetate is used as buffering agent with acetic acid. It is used in many areas like cosmetics, pharmaceuticals, agriculture, bronzing and textile industry. It is employed in production of dye materials, as a polymerization catalyst, as a polymer stabilizer, as a plating agent, preparation of gel stains, preservative in food production and flavor enhancer in the nutrient industry. Sodium acetate used in the study of lithography, photography. It is used as a base in several organic reactions in research laboratories and industries.
Solubility
Very soluble in water. Soluble in alcohol.
Notes
Hygroscopic. It is incompatible with strong oxidizing agents, halogens and it is moisture sensitive.
RUO – Research Use Only
General References:
- Kharat, S. J. Density, Viscosity and Ultrasonic Velocity Studies of Aqueous Solutions of Sodium Acetate at Different Temperatures. J. Mol. Liq. 2008, 140 (1-3), 10-14.
- Rajabzadeh, M. Determination of Unknown Concentrations of Sodium Acetate Using the Method of Standard Addition and Proton NMR: An Experiment for the Undergraduate Analytical Chemistry Laboratory. J. Chem. Educ. 2012, 89 (11), 1454-1457.
- Costa, C.; Conte, A.; Del Nobile, M. A. Effective preservation techniques to prolong the shelf life of ready-to-eat oysters. J. Sci. Food. Agric. 2014, 94 (13), 2661-2667.
- Li, S.; Huang, G. H.; An, C. J.; Yu, H. Effect of different buffer agents on in-vessel composting of food waste performance analysis and comparative study. J. Environ. Sci. Health A Tox Hazard. Subst. Environ. Eng. 2013, 48 (7), 772-780.