Search
Thermo Scientific Chemicals
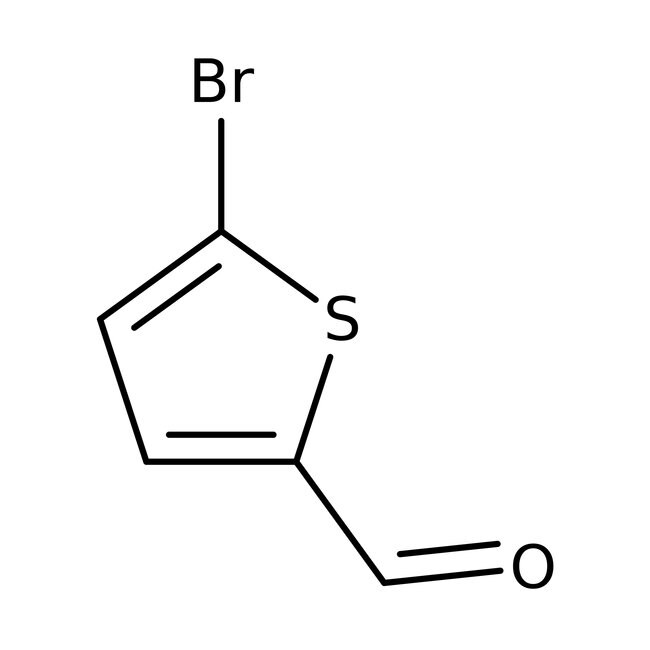
5-Bromothiophene-2-carboxaldehyde, 97%
CAS: 4701-17-1 | C5H3BrOS | 191.04 g/mol
化学物質識別子
CAS4701-17-1
IUPAC Name5-bromothiophene-2-carbaldehyde
Molecular FormulaC5H3BrOS
InChI KeyGFBVUFQNHLUCPX-UHFFFAOYSA-N
SMILESBrC1=CC=C(S1)C=O
さらに表示
仕様 スペックシート
スペックシート
Appearance (Color)Clear, pale yellow to yellow to orange or brown to dark brown
Assay (GC)>96.0%
Free acid (titration)<1.0%
Refractive Index1.6365-1.6415 @ 20?C
FormLiquid
5-Bromo-2-thiophenecarboxaldehyde was used to prepare 5-[18F]fluoro-2-2-thiophene carboxaldehyde. Also is used in biological studies as anti-inflammatory and anti-tumor activity of the marine mangrove Rhizophora apiculata.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
5-Bromo-2-thiophenecarboxaldehyde was used to prepare 5-[18F]fluoro-2-2-thiophene carboxaldehyde. Also is used in biological studies as anti-inflammatory and anti-tumor activity of the marine mangrove Rhizophora apiculata.
Solubility
Soluble in chloroform and methanol.
Notes
Air Sensitive, avoid contact with air/oxygen. Store in cool, dry place in tightly closed container.
5-Bromo-2-thiophenecarboxaldehyde was used to prepare 5-[18F]fluoro-2-2-thiophene carboxaldehyde. Also is used in biological studies as anti-inflammatory and anti-tumor activity of the marine mangrove Rhizophora apiculata.
Solubility
Soluble in chloroform and methanol.
Notes
Air Sensitive, avoid contact with air/oxygen. Store in cool, dry place in tightly closed container.
RUO – Research Use Only
General References:
- M R Kilbourn. Thiophenes as phenyl bio-isosteres: application in radiopharmaceutical design--I. Dopamine uptake antagonists. International Journal of Radiation Applications and Instrumentation. Part B. Nuclear Medicine and Biology. 1989, 16 (7), 681-686.
- V. Vinod Prabhua; C. Guruvayoorappan. Anti-inflammatory and anti-tumor activity of the marine mangrove Rhizophora apiculata. Journal of Immunotoxicology, 2012, 9 (4), 341-352.
- Starting material for synthesis of bithiophene chromophores for nonlinear optical materials: J. Org. Chem., 61, 2242 (1996).