Search
Thermo Scientific Chemicals
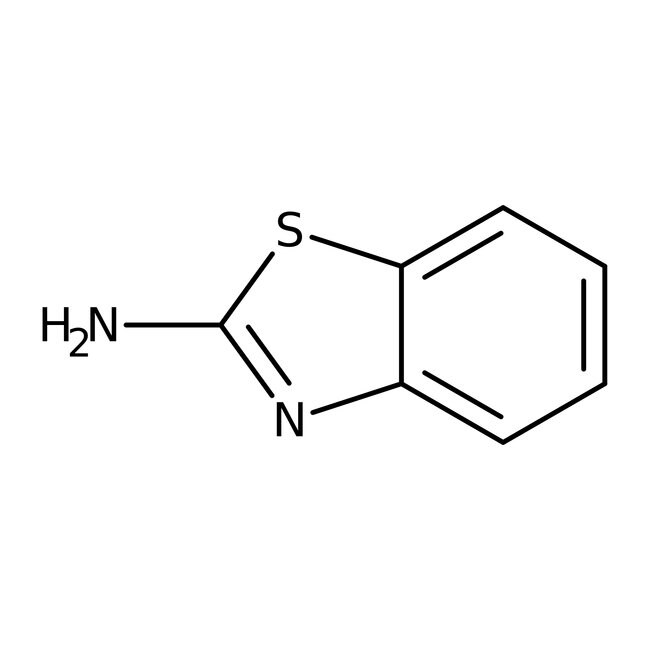
2-Aminobenzothiazole, 98%
CAS: 136-95-8 | C7H6N2S | 150.199 g/mol
化学物質識別子
CAS136-95-8
IUPAC Name1,3-benzothiazol-2-amine
Molecular FormulaC7H6N2S
InChI KeyUHGULLIUJBCTEF-UHFFFAOYSA-N
SMILESNC1=NC2=CC=CC=C2S1
さらに表示
仕様 スペックシート
スペックシート
FormPowder
Appearance (Color)White to cream
Assay (GC)≥97.5%
Assay (Non-aqueous acid-base Titration)≥97.5 to ≤102.5%
2-Aminobenzothiazole was used in the synthesis of cobalt(II) picrate mixed-ligand complexes. It was used to study adsorption of biologically significant 2-aminobenzothiazole molecules on colloidal silver particles using surface-enhanced raman scattering spectroscopy
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
2-Aminobenzothiazole was used in the synthesis of cobalt(II) picrate mixed-ligand complexes. It was used to study adsorption of biologically significant 2-aminobenzothiazole molecules on colloidal silver particles using surface-enhanced raman scattering spectroscopy
Solubility
Soluble in alcohol, chloroform, diethyl ether.
Notes
Store away from strong oxidizing agents. Keep container tightly closed. Store in cool, dry conditions in well sealed containers.
2-Aminobenzothiazole was used in the synthesis of cobalt(II) picrate mixed-ligand complexes. It was used to study adsorption of biologically significant 2-aminobenzothiazole molecules on colloidal silver particles using surface-enhanced raman scattering spectroscopy
Solubility
Soluble in alcohol, chloroform, diethyl ether.
Notes
Store away from strong oxidizing agents. Keep container tightly closed. Store in cool, dry conditions in well sealed containers.
RUO – Research Use Only
General References:
- Qiuping Ding.; Xiaodan He.; Jie Wu. Synthesis of 2-Aminobenzothiazole via Copper(I)-Catalyzed Tandem Reaction of 2-Iodobenzenamine with Isothiocyanate. J. Comb. Chem. 2009, 11, (14), 587-591.
- Morteza Akhond.; Mohammad Bagher Najafi.; Javad Tashkhourian. A new cerium (III)-selective membrane electrode based on 2-aminobenzothiazole. Sensors and Actuators B: Chemical. 2004, 99, (2-3), 410-415.