Search
Thermo Scientific Chemicals
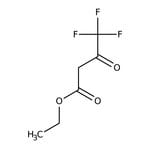
Ethyl trifluoroacetoacetate, 97%
CAS: 372-31-6 | C6H7F3O3 | 184.114 g/mol
化学物質識別子
CAS372-31-6
IUPAC Nameethyl 4,4,4-trifluoro-3-oxobutanoate
Molecular FormulaC6H7F3O3
InChI KeyOCJKUQIPRNZDTK-UHFFFAOYSA-N
SMILESCCOC(=O)CC(=O)C(F)(F)F
さらに表示
仕様 スペックシート
スペックシート
FormLiquid
Appearance (Color)Clear colorless to pale yellow
Assay (GC)≥96.0%
Refractive Index1.3750-1.3790 @ 20?C
Ethyl trifluoroacetoacetate is used as an intermediate in synthetic chemistry. It is also used as an intermediate for agro chemicals and pharmaceuticals. Further, it is used in the preparation of fluorinated 2-thiouracil analogs as antithyroid agents.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Ethyl trifluoroacetoacetate is used as an intermediate in synthetic chemistry. It is also used as an intermediate for agro chemicals and pharmaceuticals. Further, it is used in the preparation of fluorinated 2-thiouracil analogs as antithyroid agents.
Solubility
Miscible with water, ethanol, benzene and organic solvents.
Notes
Incompatible with oxidizing agents, strong acids, strong bases and strong reducing agents.
Ethyl trifluoroacetoacetate is used as an intermediate in synthetic chemistry. It is also used as an intermediate for agro chemicals and pharmaceuticals. Further, it is used in the preparation of fluorinated 2-thiouracil analogs as antithyroid agents.
Solubility
Miscible with water, ethanol, benzene and organic solvents.
Notes
Incompatible with oxidizing agents, strong acids, strong bases and strong reducing agents.
RUO – Research Use Only
General References:
- For discussion of factors influencing O- vs C-alkylation, see: J. Chem. Soc., Chem. Commun., 83 (1989).
- Mogilaiah, K.; Rao, A. N.; Rao, P. K. ZrOCl2.8H2O catalyzed solvent-free Friedlander synthesis of 1,8-naphthyridines. Indian J. Chem. 2015, 54 (10), 1280-1282.
- Lensen, N.; Marais, J.; Brigaud, T. Straightforward Synthesis of Novel Enantiopure alpha-Trifluoromethylated Azetidine 2-Carboxylic Acid and Homoserines. Org. Lett. 2015, 17 (2), 342-345.