Search
Thermo Scientific Chemicals
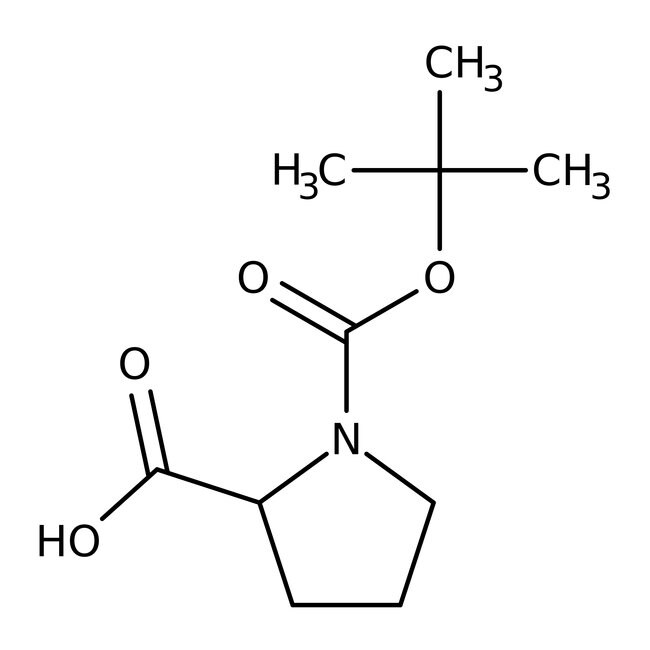
N-Boc-L-proline, 99%, Thermo Scientific Chemicals
化学物質識別子
CAS15761-39-4
IUPAC Name1-[(tert-butoxy)carbonyl]pyrrolidine-2-carboxylic acid
Molecular FormulaC10H17NO4
InChI KeyZQEBQGAAWMOMAI-UHFFFAOYNA-N
SMILESCC(C)(C)OC(=O)N1CCCC1C(O)=O
さらに表示
仕様 スペックシート
スペックシート
FormCrystals or powder or crystalline powder
Melting Point (clear melt)131.0-137.0°C
Appearance (Color)White
Assay (HPLC)≥98.5%
Assay (Aqueous acid-base Titration)≥98.5 to ≤101.5%
さらに表示
N-Boc-L-proline is used as an intermediate in organic synthesis. It is also used to prepare daclatasvir, which inhibits the hepatitis C virus (HCV) non-structural 5A (NS5A) protein. Further, it is used as a drug in the treatment of hepatitis C virus (HCV).
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
N-Boc-L-proline is used as an intermediate in organic synthesis. It is also used to prepare daclatasvir, which inhibits the hepatitis C virus (HCV) non-structural 5A (NS5A) protein. Further, it is used as a drug in the treatment of hepatitis C virus (HCV).
Solubility
Soluble in acetic acid.
Notes
Moisture and light sensitive. Incompatible with strong oxidizing agents.
N-Boc-L-proline is used as an intermediate in organic synthesis. It is also used to prepare daclatasvir, which inhibits the hepatitis C virus (HCV) non-structural 5A (NS5A) protein. Further, it is used as a drug in the treatment of hepatitis C virus (HCV).
Solubility
Soluble in acetic acid.
Notes
Moisture and light sensitive. Incompatible with strong oxidizing agents.
RUO – Research Use Only
General References:
- Sultane, P. R.; Mete, T. B.; Bhat, R. G. A convenient protocol for the deprotection of N-benzyloxycarbonyl (Cbz) and benzyl ester groups. Tetrahedron Lett. 2015, 56 (16), 2067-2070.
- Wang, H.; Li, N.; Yan, Z.; Zhang, J.; Wan, X. Synthesis and properties of novel helical 3-vinylpyridine polymers containing proline moieties for asymmetric aldol reaction. RSC Adv. 2015, 5 (65), 52410-52419.