Search
Thermo Scientific™
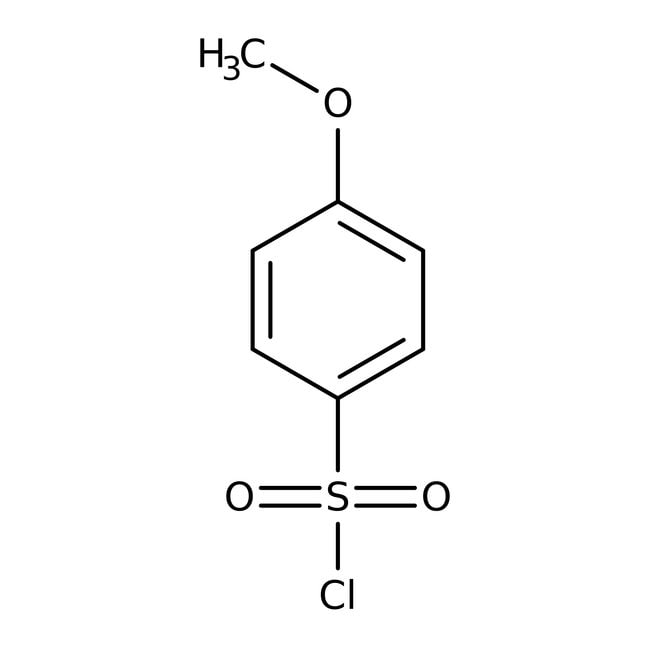
4-Methoxybenzenesulfonyl chloride, 98+%
CAS: 98-68-0 | C7H7ClO3S | 206.64 g/mol
化学物質識別子
CAS98-68-0
IUPAC Name4-methoxybenzene-1-sulfonyl chloride
Molecular FormulaC7H7ClO3S
InChI KeyDTJVECUKADWGMO-UHFFFAOYSA-N
SMILESCOC1=CC=C(C=C1)S(Cl)(=O)=O
さらに表示
仕様 スペックシート
スペックシート
Appearance (Color)White to pale cream
Assay (Titration ex Chloride)≥98.0 to ≤102.0%
FormCrystals or powder or crystalline powder or lumps or chunks or fused solid
Assay (GC)≥98.0%
Melting Point (clear melt)36.0-43.0?C
4-Methoxybenzenesulfonyl chloride is used as an anti-HIV as Pharmaceutical Intermediates. It also acts as protecting group agent for various nitrogen functions.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
4-Methoxybenzenesulfonyl chloride is used as an anti-HIV as Pharmaceutical Intermediates. It also acts as protecting group agent for various nitrogen functions.
Solubility
Soluble in Toluene. Decomposes in water.
Notes
Moisture Sensitive. Store at 4°C. Store in a cool and dark place. Protect from moisture. Store away from strong bases.
4-Methoxybenzenesulfonyl chloride is used as an anti-HIV as Pharmaceutical Intermediates. It also acts as protecting group agent for various nitrogen functions.
Solubility
Soluble in Toluene. Decomposes in water.
Notes
Moisture Sensitive. Store at 4°C. Store in a cool and dark place. Protect from moisture. Store away from strong bases.
RUO – Research Use Only
General References:
- Yoshihiko Watanabe; Nobuyuki Mase; Moto-aki Tateyama; Takeshi Toru. An improved and efficient procedure for the preparation of chiral sulfinates from sulfonyl chloride using triphenylphosphine.Tetrahedron: Asymmetry.1999, 10 737-745.
- Yujie Sun; Chengyin Wang; Qunyin Wen; Guoxiu Wang; Honghai Wang; Qishu Qu; Xiaoya Hu. Determination of Glyphosate and Aminomethylphosphonic Acid in Water by LC Using a New Labeling Reagent, 4-Methoxybenzenesulfonyl Fluoride.Chromatographia.2010, 72 679-686.
- Amines and other N-functions can be protected as 4-methoxybenzenesufonyl (PMBS, Mps) derivatives: Chem. Pharm. Bull., 29, 2592 (1981), useful for the imidazole in histidines, stable to TFA and HBr/AcOH: Bull. Chem. Soc. Jpn., 47, 3146 (1974); J. Org. Chem., 45, 547 (1980); deprotection in TFA is promoted by dimethyl sulfide: J. Chem. Soc., Chem. Commun., 955 (1979). For deprotection of PMBS-indoles using Mg in methanol, see: J. Org. Chem., 62, 6519 (1997).
- Also used for protection of the guanidino function of arginine residues in peptide synthesis. The sulfonamide is cleaved by triflic acid under less drastic conditions than tosyl groups: Chem. Pharm. Bull., 24, 1568 (1976). Compare also 4-Methoxy-2,3,6-trimethyl benzenesulfonyl chloride, L11829. See Appendix 6.