Search
Thermo Scientific Chemicals
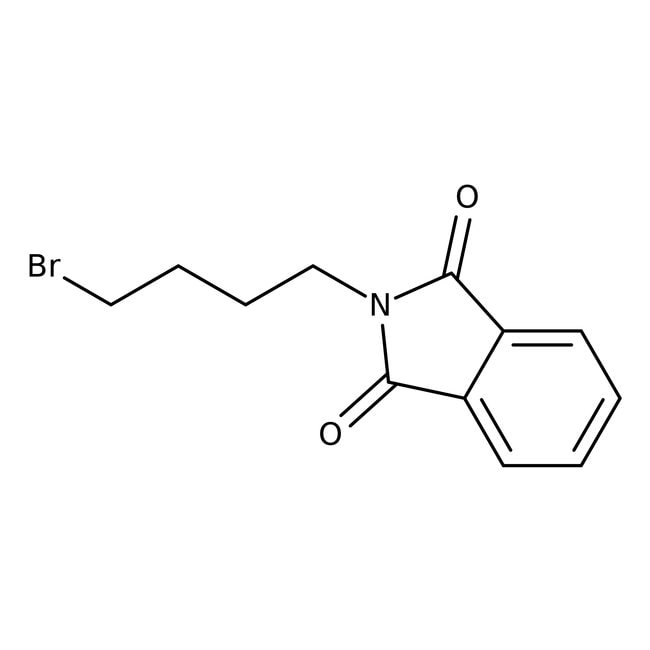
N-(4-Bromobutyl)phthalimide, 96%
CAS: 5394-18-3 | C12H12BrNO2 | 282.14 g/mol
化学物質識別子
CAS5394-18-3
N-(4-Bromobutyl)phthalimide is used in organic synthesis and the production of pharmaceutical. It can react with 1-phenyl-piperazine to get N-[4-(4-phenyl-piperazin-1-yl)-butyl]-phthalimide. It is a useful synthesis reagent used to synthesize B-cyclodextrin derivatives.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
N-(4-Bromobutyl)phthalimide is used in organic synthesis and the production of pharmaceutical. It can react with 1-phenyl-piperazine to get N-[4-(4-phenyl-piperazin-1-yl)-butyl]-phthalimide. It is a useful synthesis reagent used to synthesize B-cyclodextrin derivatives.
Solubility
Soluble in ethanol. Insoluble in water.
Notes
Store at room temperature. Store away from oxidizing agents.
N-(4-Bromobutyl)phthalimide is used in organic synthesis and the production of pharmaceutical. It can react with 1-phenyl-piperazine to get N-[4-(4-phenyl-piperazin-1-yl)-butyl]-phthalimide. It is a useful synthesis reagent used to synthesize B-cyclodextrin derivatives.
Solubility
Soluble in ethanol. Insoluble in water.
Notes
Store at room temperature. Store away from oxidizing agents.
RUO – Research Use Only
General References:
- S. Tomasi; M. Le Roch; J. Renault; J. C. Corbel and P. Uriac. N-alkylation of N1-mesitylenesulphonylputrescine with N-(4-bromobutyl)phthalimide: A Parallel Approach Using Multipin Solid-phase Synthesis. Pharmacy and Pharmacology Communications. 2000, 6 (4), 155-159.
- Richard A. Glennon; Noreen A. Naiman; M. Edward Pierson; J. Doyle Smith; Abd M. Ismaiel; Milt Titeler; Robert A. Lyon. N-(phthalimidoalkyl) derivatives of serotonergic agents: a common interaction at 5-HT1A serotonin binding sites?. J. Med. Chem. 1989, 32 (8), 1921-1926.