Search Thermo Fisher Scientific
Thermo Scientific Chemicals
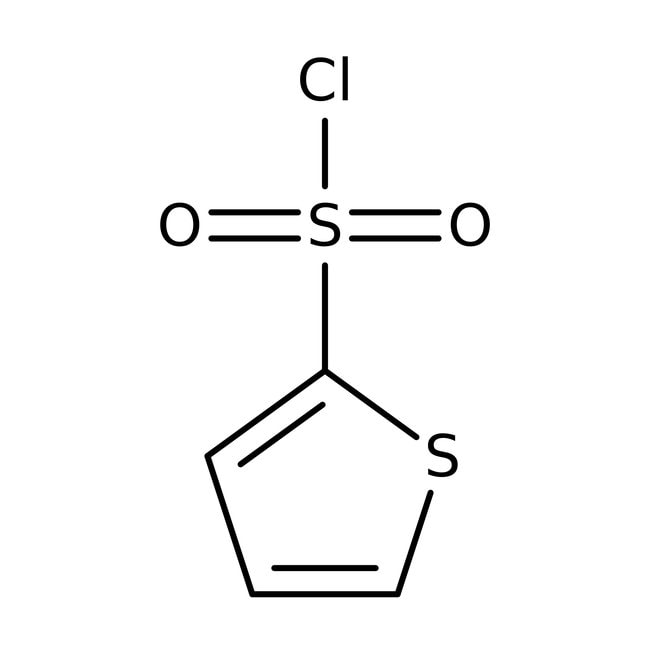
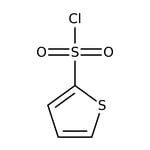
Thiophene-2-sulfonyl chloride, 97%, Thermo Scientific Chemicals
製品番号(カタログ番号): A15553.06
5 g, Each
化学物質識別子
CAS
16629-19-9
IUPAC Name
thiophene-2-sulfonyl chloride
Molecular Formula
C4H3ClO2S2
InChI Key
VNNLHYZDXIBHKZ-UHFFFAOYSA-N
SMILES
ClS(=O)(=O)C1=CC=CS1
仕様
Form
Crystals or powder or crystalline powder or chunks or fused solid
Melting Point (clear melt)
26.0-35.0?C
Appearance (Color)
White to orange or blue to pale gray to gray to black
Assay (GC)
≥96.0%
概要
2-Thiophenesulfonyl chloride has been used in the preparation of 2-((trans-2-phenyl cyclopropyl)sulfonyl)thiophene
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
2-Thiophenesulfonyl chloride has been used in the preparation of 2-((trans-2-phenyl cyclopropyl)sulfonyl)thiophene
Solubility
Reacts with water.
Notes
Moisture sensitive. Store under inert gas. Store away from moisture, oxidizing agents, bases.
2-Thiophenesulfonyl chloride has been used in the preparation of 2-((trans-2-phenyl cyclopropyl)sulfonyl)thiophene
Solubility
Reacts with water.
Notes
Moisture sensitive. Store under inert gas. Store away from moisture, oxidizing agents, bases.
RUO – Research Use Only
図
ドキュメントおよびダウンロード
証明書
ロット番号または部分ロット番号で検索
よくあるご質問(FAQ)
引用および参考文献
Search citations by name, author, journal title or abstract text