Search
Thermo Scientific Chemicals
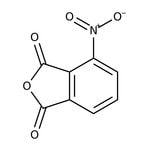
3-Nitrophthalic anhydride, 97%
CAS: 641-70-3 | C8H3NO5 | 193.11 g/mol
化学物質識別子
CAS641-70-3
IUPAC Name4-nitro-1,3-dihydro-2-benzofuran-1,3-dione
Molecular FormulaC8H3NO5
InChI KeyROFZMKDROVBLNY-UHFFFAOYSA-N
SMILES[O-][N+](=O)C1=CC=CC2=C1C(=O)OC2=O
さらに表示
仕様 スペックシート
スペックシート
Melting Point (clear melt)160-166°C
Appearance (Color)White to yellow to pale brown or cream
FormCrystalline powder
Assay (Aqueous acid-base Titration)≥96.0%
4-Nitrophthalic Anhydride is an intermediate for the synthesis of the benzimidazole PARP inhibitor I, ABT-472. It reacts with aminoquinazolinones to yield phthalimidoquinazolinones.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
4-Nitrophthalic Anhydride is an intermediate for the synthesis of the benzimidazole PARP inhibitor I, ABT-472. It reacts with aminoquinazolinones to yield phthalimidoquinazolinones.
Solubility
May decompose in water.
Notes
Air sensitive. Store at room temperature. Keep away from oxidizing agents.
4-Nitrophthalic Anhydride is an intermediate for the synthesis of the benzimidazole PARP inhibitor I, ABT-472. It reacts with aminoquinazolinones to yield phthalimidoquinazolinones.
Solubility
May decompose in water.
Notes
Air sensitive. Store at room temperature. Keep away from oxidizing agents.
RUO – Research Use Only
General References:
- Ben H. Nicolet.; Jacob Sacks.3-Nitrophthalic Anhydride As A Reagent For Alcohols.J. Am. Chem. Soc. 1925, 47 (9),2348-2350 .
- E. Wertheim. Derivatives For The Identification Of Mercaptans. J. Am. Chem. Soc. 1929, 51 (12),3661-3664 .
- Has been used as an N-blocking group for amino acids in solid-phase peptide synthesis, providing an acidic 'handle' allowing separation by ion-exchange chromatography: Angew. Chem. Int. Ed., 8, 764 (1969). See Appendix 6.