Search
Thermo Scientific Chemicals
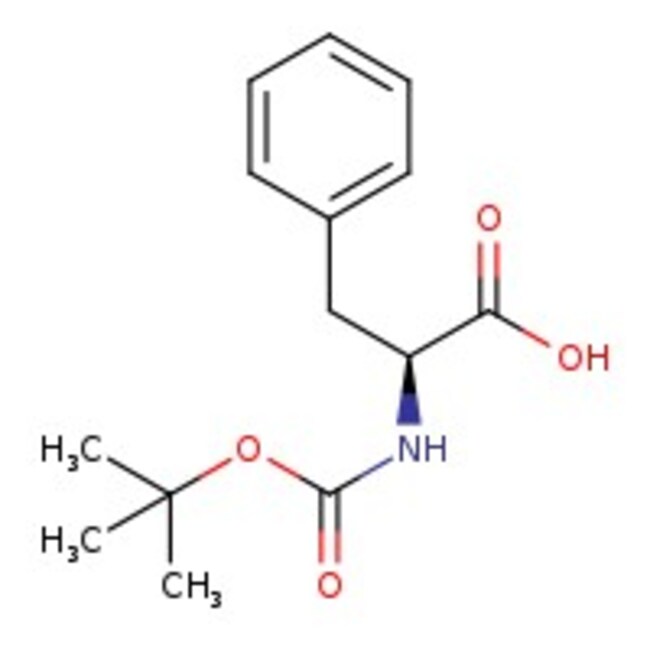
N-Boc-L-phenylalanine, 99%
CAS: 13734-34-4 | C14H19NO4 | 265.31 g/mol
化学物質識別子
CAS13734-34-4
IUPAC Name(2S)-2-{[(tert-butoxy)carbonyl]amino}-3-phenylpropanoic acid
Molecular FormulaC14H19NO4
InChI KeyZYJPUMXJBDHSIF-NSHDSACASA-N
SMILESCC(C)(C)OC(=O)N[C@@H](CC1=CC=CC=C1)C(O)=O
さらに表示
仕様 スペックシート
スペックシート
Appearance (Color)White
Assay (Aqueous acid-base Titration)≥98.5 to ≤101.5%
Melting Point (clear melt)83.0-89.0?C
FormCrystals or powder or crystalline powder
Optical Rotation24.4 ± 0.1? (C=1 in ethanol)
N-Boc-L-phenylalanine is a derivative of Phenylalanine used in enantioselective hydrolysis of amino acid esters.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
N-Boc-L-phenylalanine is a derivative of Phenylalanine used in enantioselective hydrolysis of amino acid esters.
Solubility
Soluble in methanol, dichloromethane, dimethylformamide and N-methyl-2-pyrrolidone.
Notes
Keep container tightly sealed. Store in cool, dry conditions in well sealed containers. Incompatible with oxidizing agents.
N-Boc-L-phenylalanine is a derivative of Phenylalanine used in enantioselective hydrolysis of amino acid esters.
Solubility
Soluble in methanol, dichloromethane, dimethylformamide and N-methyl-2-pyrrolidone.
Notes
Keep container tightly sealed. Store in cool, dry conditions in well sealed containers. Incompatible with oxidizing agents.
RUO – Research Use Only
General References:
- Marie Claude Fournie-Zaluski, Pascale Coric, Serge Turcaud, Luce Bruetschy, Evelyne Lucas, Florence Noble, Bernard P. Roques. Potent and systemically active aminopeptidase N inhibitors designed from active-site investigation. J. Med. Chem. 1992, 35 (7), 1259-1266.
- Sung-Hwan Moon, Yasufumi Ohfune. Efficient Syntheses of the Four Enantiomers and Diastereomers of .alpha.-Methylthreonine and Both Enantiomers of .alpha.-Methylserine. J. Am. Chem. Soc. 1994, 116 (16), 7405-7406.