Search
Thermo Scientific Chemicals
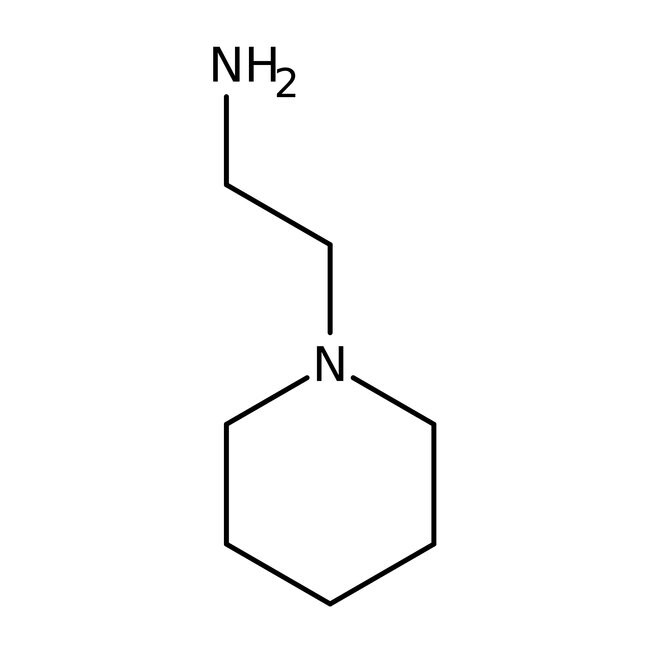
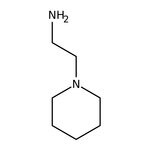
1-(2-Aminoethyl)piperidine, 98%
CAS: 27578-60-5 | C7H16N2 | 128.219 g/mol
化学物質識別子
CAS27578-60-5
IUPAC Name2-(piperidin-1-yl)ethan-1-amine
Molecular FormulaC7H16N2
InChI KeyCJNRGSHEMCMUOE-UHFFFAOYSA-N
SMILESNCCN1CCCCC1
さらに表示
仕様 スペックシート
スペックシート
Appearance (Color)Clear colorless to pale yellow
Assay (GC)≥97.5%
Refractive Index1.4725-1.4765 @ 20?C
FormLiquid
1-(2-Aminoethyl)piperidine is used in modification of vinylbenzyl chloride/divinylbenzene gel copolymer bead and in the synthesis of linkage isomers trans-bis[1-(2-aminoethyl)piperidine]dinitronickel and trans-bis[1-(2-aminoethyl)-piperidine]dinitritonickel. It is also used as a reactant for synthesis of: analogs of anticancer agents, inhibitor of botulinum neurotoxin serotype A light chain, P. falciparum malaria, and Ebola filovirus, cannabinoid CB1 receptor antagonists, small molecules that restore E-cadherin expression and reduce invasion in colorectal carcinoma cells, potent and selective 5-HT6 antagonists and N-mustards as anticancer agents. 1-(2-Aminoethyl)piperidine on [1+1] condensation reaction with 3-methoxy salicylaldehyde yields tridentate Schiff base ligand.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
1-(2-Aminoethyl)piperidine is used in modification of vinylbenzyl chloride/divinylbenzene gel copolymer bead and in the synthesis of linkage isomers trans-bis[1-(2-aminoethyl)piperidine]dinitronickel and trans-bis[1-(2-aminoethyl)-piperidine]dinitritonickel. It is also used as a reactant for synthesis of: analogs of anticancer agents, inhibitor of botulinum neurotoxin serotype A light chain, P. falciparum malaria, and Ebola filovirus, cannabinoid CB1 receptor antagonists, small molecules that restore E-cadherin expression and reduce invasion in colorectal carcinoma cells, potent and selective 5-HT6 antagonists and N-mustards as anticancer agents. 1-(2-Aminoethyl)piperidine on [1+1] condensation reaction with 3-methoxy salicylaldehyde yields tridentate Schiff base ligand.
Solubility
Partly miscible in water.
Notes
Keep container tightly sealed. Store in cool, dry conditions in well sealed containers. Incompatible with oxidizing agents, acid chlorides and acid anhydrides. It is sensitive to air.
1-(2-Aminoethyl)piperidine is used in modification of vinylbenzyl chloride/divinylbenzene gel copolymer bead and in the synthesis of linkage isomers trans-bis[1-(2-aminoethyl)piperidine]dinitronickel and trans-bis[1-(2-aminoethyl)-piperidine]dinitritonickel. It is also used as a reactant for synthesis of: analogs of anticancer agents, inhibitor of botulinum neurotoxin serotype A light chain, P. falciparum malaria, and Ebola filovirus, cannabinoid CB1 receptor antagonists, small molecules that restore E-cadherin expression and reduce invasion in colorectal carcinoma cells, potent and selective 5-HT6 antagonists and N-mustards as anticancer agents. 1-(2-Aminoethyl)piperidine on [1+1] condensation reaction with 3-methoxy salicylaldehyde yields tridentate Schiff base ligand.
Solubility
Partly miscible in water.
Notes
Keep container tightly sealed. Store in cool, dry conditions in well sealed containers. Incompatible with oxidizing agents, acid chlorides and acid anhydrides. It is sensitive to air.
RUO – Research Use Only
General References:
- Dorota Jermakowicz-Bartkowiak. Polymer resins for recovery of valuable metals. Environmental Geochemistry and Health. 2010, 32 (4), 317-320.
- Sujit Sasmal.; Sohini Sarkar.; Núria Aliaga-Alcalde.; Sasankasekhar Mohanta. Syntheses, structures, and magnetic properties of three one-dimensional end-to-end azide/cyanate-bridged copper(II) compounds exhibiting ferromagnetic interaction: new type of solid state isomerism. Inorganic Chemistry. 2011, 50 (12), 5687-5695.