Search
Thermo Scientific Chemicals
Sodium nitrite, 98%
CAS: 7632-00-0 | NaNO2 | 69.00 g/mol
化学物質識別子
CAS7632-00-0
IUPAC Namesodium nitrite
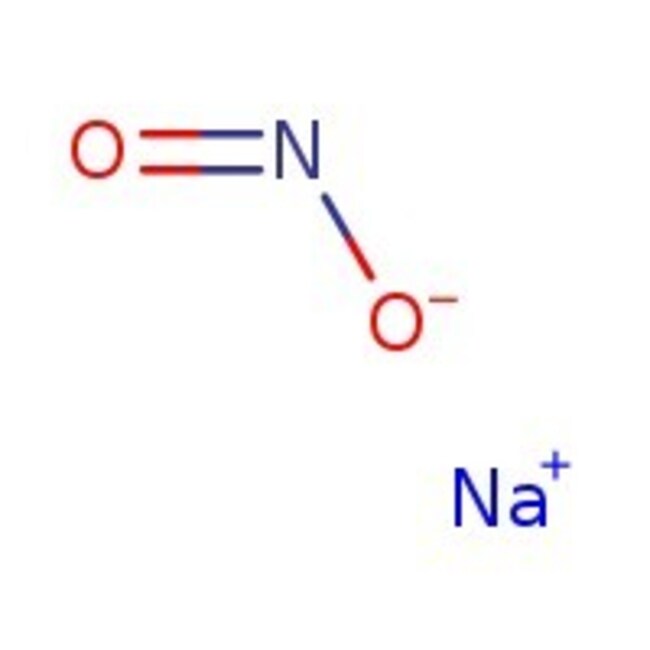
Molecular FormulaNNaO2
InChI KeyLPXPTNMVRIOKMN-UHFFFAOYSA-M
SMILES[Na+].[O-]N=O
さらに表示
仕様 スペックシート
スペックシート
Insoluble materialIn water: ≤0.05%w/w (US sourced material)
Melting Point278-283?C (UK sourced material)
Appearance (Color)White to cream or pale yellow (UK and US sourced material)
Assay (Redox Titration)≥97.5 to ≤102.5% (UK sourced material)
Identification (FTIR)Conforms (UK sourced material)
さらに表示
Sodium nitrite is used in many industrial applications viz. in meat curing, coloring and preserving. It is used as a reagent in analytical chemistry, an antidote in cyanide poisoning, an electrolyte in electrochemical grinding, as a cooling solution in closed loop systems and as an additive in industrial greases. It finds application as a corrosion inhibitor as well as in the rubber industry. In metallurgy, it is used for phosphatizing and detinning. It acts as a precursor to diazo dyes, nitroso compounds and various organic compounds like pharmaceuticals. As a food additive, it is used to prevent botulism.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Sodium nitrite is used in many industrial applications viz. in meat curing, coloring and preserving. It is used as a reagent in analytical chemistry, an antidote in cyanide poisoning, an electrolyte in electrochemical grinding, as a cooling solution in closed loop systems and as an additive in industrial greases. It finds application as a corrosion inhibitor as well as in the rubber industry. In metallurgy, it is used for phosphatizing and detinning. It acts as a precursor to diazo dyes, nitroso compounds and various organic compounds like pharmaceuticals. As a food additive, it is used to prevent botulism.
Solubility
Soluble in water, methanol, ethanol, ammonia, nitric acid. Slightly soluble diethyl ether.
Notes
Decomposes above 320°. Hygroscopic. Incompatible with reducing agents, strong oxidizing agents, organics, finely powdered metals.
Sodium nitrite is used in many industrial applications viz. in meat curing, coloring and preserving. It is used as a reagent in analytical chemistry, an antidote in cyanide poisoning, an electrolyte in electrochemical grinding, as a cooling solution in closed loop systems and as an additive in industrial greases. It finds application as a corrosion inhibitor as well as in the rubber industry. In metallurgy, it is used for phosphatizing and detinning. It acts as a precursor to diazo dyes, nitroso compounds and various organic compounds like pharmaceuticals. As a food additive, it is used to prevent botulism.
Solubility
Soluble in water, methanol, ethanol, ammonia, nitric acid. Slightly soluble diethyl ether.
Notes
Decomposes above 320°. Hygroscopic. Incompatible with reducing agents, strong oxidizing agents, organics, finely powdered metals.
RUO – Research Use Only
General References:
- Cheng, D.; Yuan, K.; Xu, X.; Yan, J. The oxidative coupling of benzylic compounds catalyzed by 2, 3-dichloro-5, 6-dicyano-benzoquinone and sodium nitrite using molecular oxygen as a co-oxidant. Tetrahedron Lett. 2015, 56 (13), 1641-1644.
- Kroflič, A.; Grilc, M.; Grgić, I. Unraveling Pathways of Guaiacol Nitration in Atmospheric Waters: Nitrite, A Source of Reactive Nitronium Ion in the Atmosphere. Environ. Sci. Technol. 2015, 49 (15), 9150-9158.