Search
Thermo Scientific Chemicals
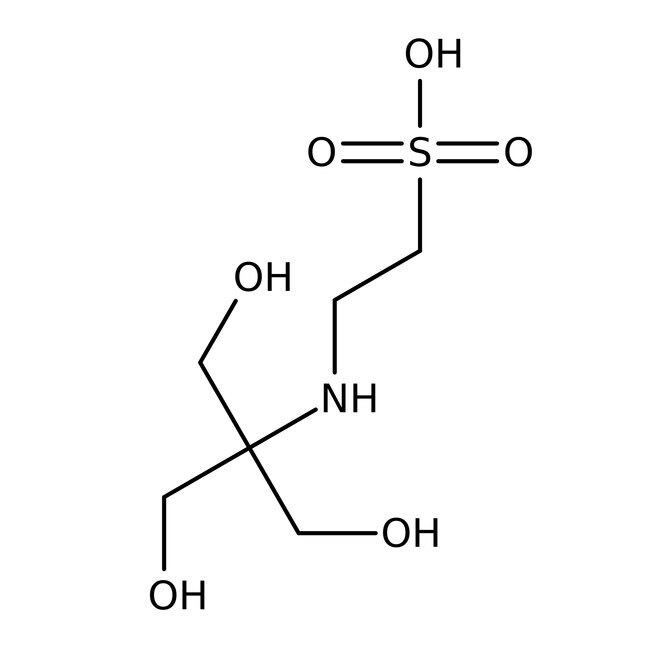
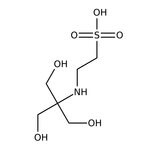
TES, 99%
CAS: 7365-44-8 | C6H15NO6S | 229.247 g/mol
化学物質識別子
CAS7365-44-8
IUPAC Name2-{[1,3-dihydroxy-2-(hydroxymethyl)propan-2-yl]amino}ethane-1-sulfonic acid
Molecular FormulaC6H15NO6S
InChI KeyJOCBASBOOFNAJA-UHFFFAOYSA-N
SMILESOCC(CO)(CO)NCCS(O)(=O)=O
さらに表示
仕様 スペックシート
スペックシート
Appearance (Color)White
FormCrystals or powder or crystalline powder
Assay (Aqueous acid-base Titration)≥98.5 to ≤101.5%
Identification (FTIR)Conforms
TES plays an important role in production of pH buffers for a biological system. It is used to make zwitterionic buffer solutions in the pH range 6.8-8.2. It is used in molecular biology, diagnosis, cell culture, pharmacy, agrochemical and pharmaceuticals. It is also used in the study of succinate oxidation and one of the components of test yolk buffer medium used in refrigeration of semen and transport.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
TES plays an important role in production of pH buffers for a biological system. It is used to make zwitterionic buffer solutions in the pH range 6.8-8.2. It is used in molecular biology, diagnosis, cell culture, pharmacy, agrochemical and pharmaceuticals. It is also used in the study of succinate oxidation and one of the components of test yolk buffer medium used in refrigeration of semen and transport.
Solubility
Soluble in water.
Notes
Incompatible with strong oxidizing agents.
TES plays an important role in production of pH buffers for a biological system. It is used to make zwitterionic buffer solutions in the pH range 6.8-8.2. It is used in molecular biology, diagnosis, cell culture, pharmacy, agrochemical and pharmaceuticals. It is also used in the study of succinate oxidation and one of the components of test yolk buffer medium used in refrigeration of semen and transport.
Solubility
Soluble in water.
Notes
Incompatible with strong oxidizing agents.
RUO – Research Use Only
General References:
- Biological buffer useful in pH range 7.0 - 8.0: Biochemistry, 5, 467 (1966).
- Gupta, B. S.; Taha, M.; Lee, M. J. Interactions of bovine serum albumin with biological buffers, TES, TAPS, and TAPSO in aqueous solutions. Process Biochem. 2013, 48 (11), 1686-1696.
- Daub, M. E.; Prudhomme, J.; Roch, K. L.; Vanderwal, C. D. Synthesis and Potent Antimalarial Activity of Kalihinol B. J. Am. Chem. Soc. 2015, 137 (15), 4912-4915.