Search
Thermo Scientific Chemicals
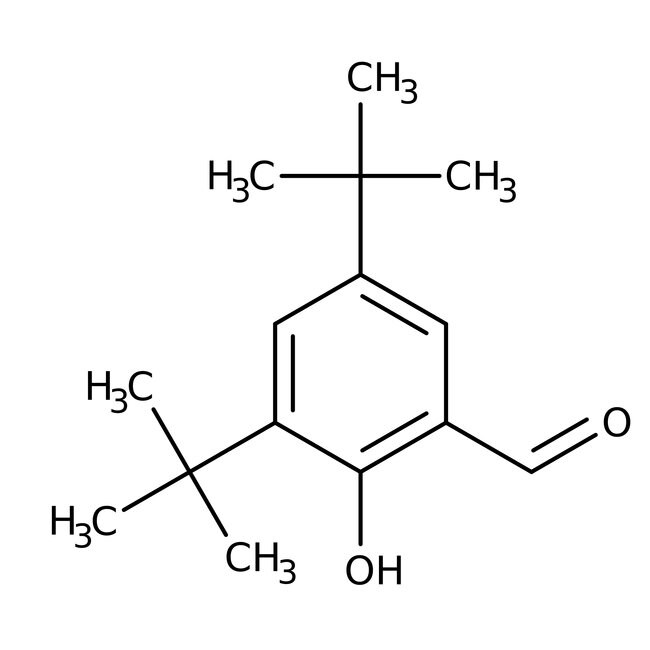
3,5-Di-tert-butyl-2-hydroxybenzaldehyde, 99%
CAS: 37942-07-7 | C15H22O2 | 234.339 g/mol
化学物質識別子
CAS37942-07-7
IUPAC Name3,5-di-tert-butyl-2-hydroxybenzaldehyde
Molecular FormulaC15H22O2
InChI KeyRRIQVLZDOZPJTH-UHFFFAOYSA-N
SMILESCC(C)(C)C1=CC(C=O)=C(O)C(=C1)C(C)(C)C
さらに表示
仕様 スペックシート
スペックシート
FormCrystalline solid or powder
Identification (FTIR)Conforms
Appearance (Color)White to cream to yellow
Assay (GC)≥98.5%
Melting Point (clear melt)58-62?C
3,5-Di-tert-butyl-2-hydroxybenzaldehyde is used in the synthesis of Mn(III)-salen complex and its diamino precursor 5,6-diamino-5,6-dideoxy-1,2-O-isopropylidene-3-O-methyl-β-L-idofuranose, chiral Schiff base ligand for an enantioselective copper-catalyzed addition of phenyl acetylene to imines, chiral oxazolidine ligand for the enantioselective addition of diethyl zinc to aldehydes and tin Schiff base complexes with histidine analogues. It has antibacterial activity and is used in the preparation nickel complexes. It is structurally related to 3,5-di-t-butylcatechol (DTCAT) but is not as potent an activator of rat skeletal muscle ryanodine receptor Ca2+ channel (RyRC).
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
3,5-Di-tert-butyl-2-hydroxybenzaldehyde is used in the synthesis of Mn(III)-salen complex and its diamino precursor 5,6-diamino-5,6-dideoxy-1,2-O-isopropylidene-3-O-methyl-β-L-idofuranose, chiral Schiff base ligand for an enantioselective copper-catalyzed addition of phenyl acetylene to imines, chiral oxazolidine ligand for the enantioselective addition of diethyl zinc to aldehydes and tin Schiff base complexes with histidine analogues. It has antibacterial activity and is used in the preparation nickel complexes. It is structurally related to 3,5-di-t-butylcatechol (DTCAT) but is not as potent an activator of rat skeletal muscle ryanodine receptor Ca2+ channel (RyRC).
Solubility
Soluble in DMSO and methanol.
Notes
Air sensitive. Store away from oxidizing agents. Keep the container tightly closed and place it in a cool, dry and well ventilated condition.
3,5-Di-tert-butyl-2-hydroxybenzaldehyde is used in the synthesis of Mn(III)-salen complex and its diamino precursor 5,6-diamino-5,6-dideoxy-1,2-O-isopropylidene-3-O-methyl-β-L-idofuranose, chiral Schiff base ligand for an enantioselective copper-catalyzed addition of phenyl acetylene to imines, chiral oxazolidine ligand for the enantioselective addition of diethyl zinc to aldehydes and tin Schiff base complexes with histidine analogues. It has antibacterial activity and is used in the preparation nickel complexes. It is structurally related to 3,5-di-t-butylcatechol (DTCAT) but is not as potent an activator of rat skeletal muscle ryanodine receptor Ca2+ channel (RyRC).
Solubility
Soluble in DMSO and methanol.
Notes
Air sensitive. Store away from oxidizing agents. Keep the container tightly closed and place it in a cool, dry and well ventilated condition.
RUO – Research Use Only
General References:
- Yan S and Klemm D. Mild and efficient synthesis of 5, 6-diamino-5, 6-dideoxy-1, 2-O-isopropylidene-3- O -methyl-ß-l-idofuranose: precursor of the first carbohydrate-derived chiral Mn (III)-salen complex. Tetrahedron.,2002,58(50), 10065-10071.
- Ariadna Garza-Ortiz, et al. Novel Organotin(IV) Schiff Base Complexes with Histidine Derivatives: Synthesis, Characterization, and Biological Activity.Bioinorg Chem Appl.,2013,2013(6), 502713.