Search
Thermo Scientific Chemicals
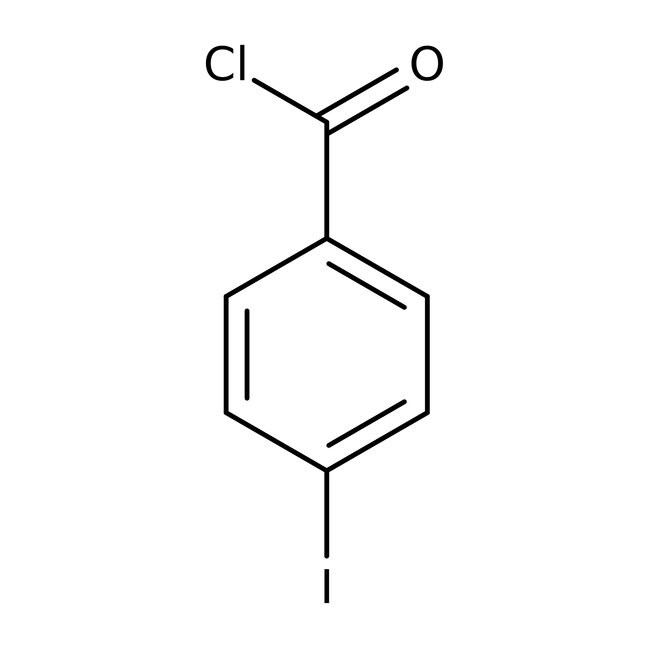
4-Iodobenzoyl chloride, 98%
CAS: 1711-02-0 | C7H4ClIO | 266.462 g/mol
化学物質識別子
CAS1711-02-0
IUPAC Name4-iodobenzoyl chloride
Molecular FormulaC7H4ClIO
InChI KeyNJAKCIUOTIPYED-UHFFFAOYSA-N
SMILESClC(=O)C1=CC=C(I)C=C1
さらに表示
仕様 スペックシート
スペックシート
Melting Point (clear melt)61.0-68.0?C
FormCrystals or powder or crystalline powder
Assay (GC)≥97.5%
Appearance (Color)White to pale cream
Assay (Titration ex Chloride)≥97.5 to ≤102.5%
4-Iodobenzoyl chloride is employed in the preparation of pyrroles by reaction with imines and acetylenes mediated by isocyanides using palladium catalysis. It is also used in the preparation of N-(1-benzylpyrrolidin-3-yl)arylbenzamides, potential human dopamine D4 antagonists. Further, it is used to prepare [2]rotaxane monomer and poly[2] rotaxane. In addition to this, it is important in the modification of poly(allylamine) in order to make the polymer X-ray visible.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
4-Iodobenzoyl chloride is employed in the preparation of pyrroles by reaction with imines and acetylenes mediated by isocyanides using palladium catalysis. It is also used in the preparation of N-(1-benzylpyrrolidin-3-yl)arylbenzamides, potential human dopamine D4 antagonists. Further, it is used to prepare [2]rotaxane monomer and poly[2] rotaxane. In addition to this, it is important in the modification of poly(allylamine) in order to make the polymer X-ray visible.
Solubility
Soluble in toluene and benzene.
Notes
Moisture sensitive. Incompatible with strong bases and water.
4-Iodobenzoyl chloride is employed in the preparation of pyrroles by reaction with imines and acetylenes mediated by isocyanides using palladium catalysis. It is also used in the preparation of N-(1-benzylpyrrolidin-3-yl)arylbenzamides, potential human dopamine D4 antagonists. Further, it is used to prepare [2]rotaxane monomer and poly[2] rotaxane. In addition to this, it is important in the modification of poly(allylamine) in order to make the polymer X-ray visible.
Solubility
Soluble in toluene and benzene.
Notes
Moisture sensitive. Incompatible with strong bases and water.
RUO – Research Use Only
General References:
- Takahashi, Y.; Miura, Y.; Yoshioka, N. Introduction of Three Aryl Groups to Benzotriazinyl Radical by Suzuki-Miyaura Cross-coupling Reaction. Chem. Lett. 2014, 43 (8), 1236-1238.
- Wang, Q.; Qian, K.; Liu, S.; Yang, Y.; Liang, B.; Zheng, C.; Yang, X.; Xu, H.; Shen, A. Q. X-ray Visible and Uniform Alginate Microspheres Loaded with in SituSynthesized BaSO4 Nanoparticles for in Vivo Transcatheter Arterial Embolization. Biomacromolecules 2015, 16 (4), 1240-1246.