Search
Thermo Scientific Chemicals
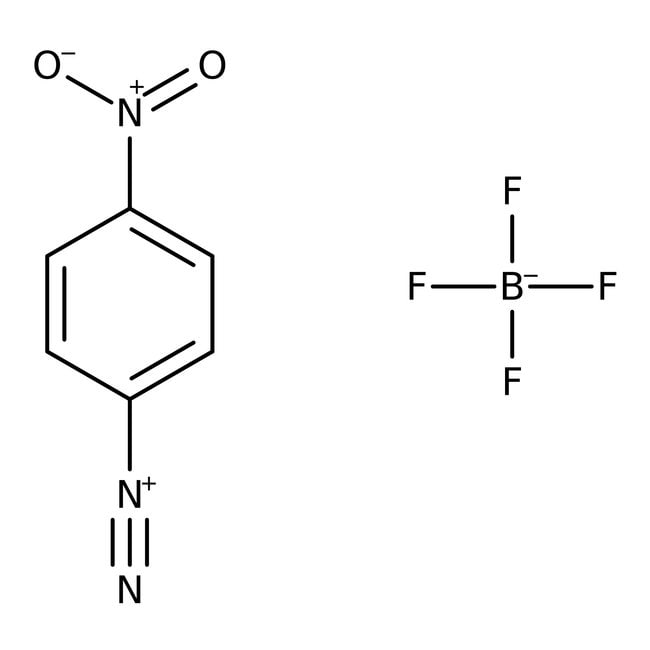
4-Nitrobenzenediazonium tetrafluoroborate, 97%
CAS: 456-27-9 | C6H4BF4N3O2 | 236.92 g/mol
化学物質識別子
CAS456-27-9
IUPAC Name4-nitrobenzene-1-diazonium; tetrafluoroboranuide
Molecular FormulaC6H4BF4N3O2
InChI KeyAFULQCYCQAHYIP-UHFFFAOYSA-N
SMILESF[B-](F)(F)F.[O-][N+](=O)C1=CC=C(C=C1)[N+]#N
さらに表示
仕様 スペックシート
スペックシート
Assay (HPLC)>96.0%
4-Nitrobenzenediazonium tetrafluoroborate is widely used as a standard reagent in organic synthesis. It is used as a dye for enterochromaffin cells. It is used for the spectrophotometric determination of common aromatic diisocyanates [toluene-2,4-diyl diisocyanate and 4,4'-methylenebis(phenyl isocyanate)] and gallic acid. It is also used for tissue staining and determination of trace phenols.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
4-Nitrobenzenediazonium tetrafluoroborate is widely used as a standard reagent in organic synthesis. It is used as a dye for enterochromaffin cells. It is used for the spectrophotometric determination of common aromatic diisocyanates [toluene-2,4-diyl diisocyanate and 4,4′-methylenebis(phenyl isocyanate)] and gallic acid. It is also used for tissue staining and determination of trace phenols.
Solubility
Soluble in acetonitrile and dimethyl sulfoxide.
Notes
Moisture sensitive. Store in a cool place. Keep the container tightly closed. Incompatible with strong bases, strong oxidizing agents and moisture.
4-Nitrobenzenediazonium tetrafluoroborate is widely used as a standard reagent in organic synthesis. It is used as a dye for enterochromaffin cells. It is used for the spectrophotometric determination of common aromatic diisocyanates [toluene-2,4-diyl diisocyanate and 4,4′-methylenebis(phenyl isocyanate)] and gallic acid. It is also used for tissue staining and determination of trace phenols.
Solubility
Soluble in acetonitrile and dimethyl sulfoxide.
Notes
Moisture sensitive. Store in a cool place. Keep the container tightly closed. Incompatible with strong bases, strong oxidizing agents and moisture.
RUO – Research Use Only
General References:
- Brisset, F.; Vieillard, J.; Berton, B.; Grognet, S. M.; Poc, C. D.; Derf, F. L. Surface functionalization of cyclic olefin copolymer with aryldiazonium salts: A covalent grafting method. Appl. Surf. Sci. 2015, 329, 337-346.
- Mooste, M.; Kibena, E.; Kozlova, J.; Marandi, M.; Matisen, L.; Niilisk, A.; Sammelselg, V.; Tammeveski, K. Electrografting and morphological studies of chemical vapour deposition grown graphene sheets modified by electroreduction of aryldiazonium salts. Electrochim. Acta 2015, 161, 195-204.