Search
Thermo Scientific Chemicals
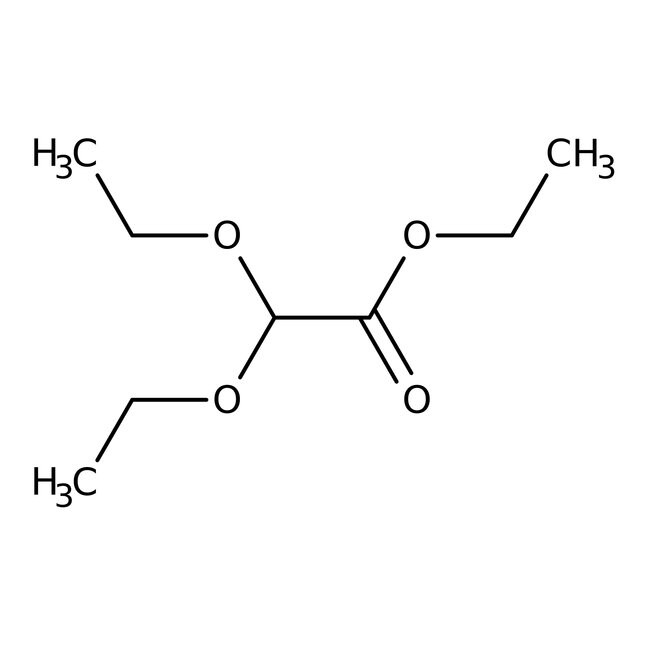
Ethyl diethoxyacetate, 97+%
CAS: 6065-82-3 | C8H16O4 | 176.212 g/mol
化学物質識別子
CAS6065-82-3
IUPAC Nameethyl 2,2-diethoxyacetate
Molecular FormulaC8H16O4
InChI KeyXCLBIKIQSCTANZ-UHFFFAOYSA-N
SMILESCCOC(OCC)C(=O)OCC
さらに表示
仕様 スペックシート
スペックシート
Refractive Index1.4080-1.4130 @ 20?C
FormLiquid
Appearance (Color)Clear colorless
Assay (GC)≥97.0%
Ethyl diethoxyacetate is used in the preparation of a fatty acid analog containing DTS, 12,13-dioxotetradecanoic acid di(N-methyl-thiosemicarbazone) (FA-DTS). It is used for synthesis of heterocyclic compounds.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Ethyl diethoxyacetate is used in the preparation of a fatty acid analog containing DTS, 12,13-dioxotetradecanoic acid di(N-methyl-thiosemicarbazone) (FA-DTS). It is used for synthesis of heterocyclic compounds.
Solubility
Soluble in Dichloromethane, Ethyl Acetate and Methanol.
Notes
Store in cool, dry conditions in well sealed containers. Keep container tightly closed.
Ethyl diethoxyacetate is used in the preparation of a fatty acid analog containing DTS, 12,13-dioxotetradecanoic acid di(N-methyl-thiosemicarbazone) (FA-DTS). It is used for synthesis of heterocyclic compounds.
Solubility
Soluble in Dichloromethane, Ethyl Acetate and Methanol.
Notes
Store in cool, dry conditions in well sealed containers. Keep container tightly closed.
RUO – Research Use Only
General References:
- Charles Heidelberger.; Robert B. Hurlbert. The Synthesis of Oxalacetic Acid-1-C14 and Orotic Acid-6-C14 1. J. Am. Chem. Soc. 1950, 72, (10), 4704-4706.
- Fieser, L.F.; Dunn, J.T. Synthesis of Plumbagin. J. Am. Chem. Soc. 1936, 58, (4), 572-575.
- Ethyl glyoxylate equivalent. Lithiation, e.g. with lithium diethylamide, provides a useful 2-carbon fragment: Tetrahedron Lett., 4551 (1975). Reaction with the bis-Grignard from 1,4-dibromobutane gives 1-(diethoxymethyl)cyclopentanol: Canad. J. Chem., 66, 168 (1988). See also Methyl dimethoxyacetate, A12171.