Search
Thermo Scientific Chemicals
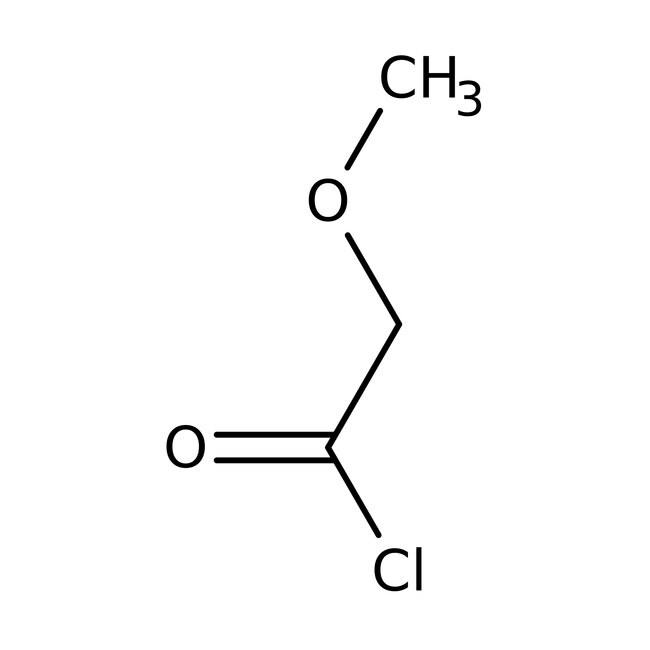
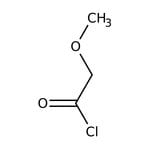
Methoxyacetyl chloride, 97%, stab. with ca 0.3% magnesium oxide
CAS: 38870-89-2 | C3H5ClO2 | 108.521 g/mol
化学物質識別子
CAS38870-89-2
IUPAC Name2-methoxyacetyl chloride
Molecular FormulaC3H5ClO2
InChI KeyJJKWHOSQTYYFAE-UHFFFAOYSA-N
SMILESCOCC(Cl)=O
さらに表示
仕様 スペックシート
スペックシート
Refractive Index1.4190 - 1.4290 @20?C
Assay (GC)> 96.0%
Formliquid with slight sediment
Appearance (Color)Colourless to pale yellow
Methoxyacetyl chloride is used as an intermediate for active pharmaceutical ingredients and dyes. Further, it acts as a precursor for the synthesis of agrochemicals.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Methoxyacetyl chloride is used as an intermediate for active pharmaceutical ingredients and dyes. Further, it acts as a precursor for the synthesis of agrochemicals.
Solubility
Miscible with acetone, ether, carbon tetrachloride and chloroform.
Notes
Moisture sensitive. Store in a cool place. Incompatible with water, alcohols, oxidizing agents and strong bases.
Methoxyacetyl chloride is used as an intermediate for active pharmaceutical ingredients and dyes. Further, it acts as a precursor for the synthesis of agrochemicals.
Solubility
Miscible with acetone, ether, carbon tetrachloride and chloroform.
Notes
Moisture sensitive. Store in a cool place. Incompatible with water, alcohols, oxidizing agents and strong bases.
RUO – Research Use Only
General References:
- Reagent for the chloromethylation of aromatics under Friedel-Crafts conditions, avoiding the use of chloromethyl methyl ether: Tetrahedron Lett., 24, 1933 (1983). For a study of the kinetics of chloromethylation of benzene and toluene in the presence of SnCl4, see: J. Org. Chem., 62, 2694 (1997). Compare Methoxyacetic acid, B22917.
- Has also been used for the protection of alcohols as methoxyacetate esters, formed in the presence of pyridine, and cleaved using methanolic or aqueous ammonia ca. 20x faster than acetate: Tetrahedron Lett., 4273 (1968). Selective cleavage with Yb(OTf)3: J. Org. Chem., 61, 4491 (1996), or ethanolamine in IPA: J. Org. Chem., 60, 331 (1995), have also been reported.
- Tiwari, D. K.; Pogula, J.; Tiwari, D. K. A general and practical route to 4,5-disubstituted oxazoles using acid chlorides and isocyanides. RSC Adv. 2015, 5 (65), 53111-53116.
- Zadehahmadi, F.; Ahmadi, F.; Tangestaninejad, S.; Moghadam, M.; Mirkhani, V.; Mohammadpoor-Baltork, I.; Kardanpour, R. Catalytic CO2 fixation using tin porphyrin supported on organic and inorganic materials under mild conditions. J. Mol. Catal. A: Chem. 2015, 398, 1-10.