Search
Thermo Scientific Chemicals
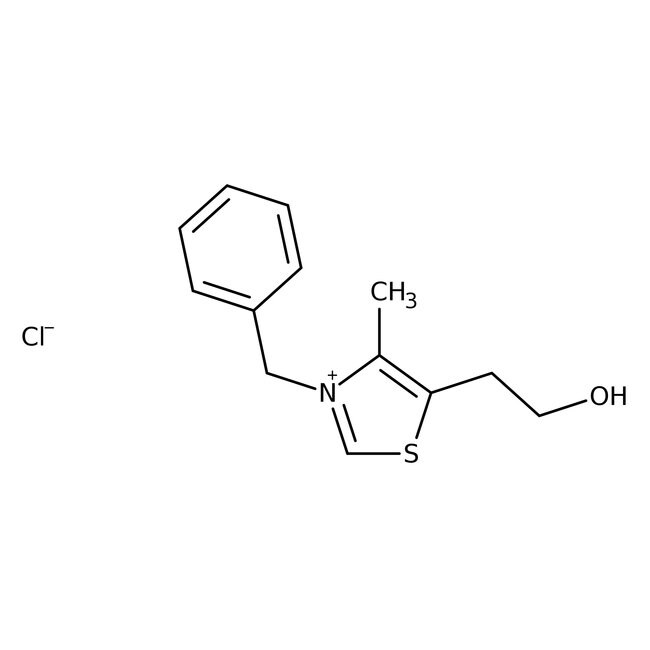
3-Benzyl-5-(2-hydroxyethyl)-4-methylthiazolium chloride, 98%
CAS: 4568-71-2 | C13H16ClNOS | 269.79 g/mol
化学物質識別子
CAS4568-71-2
IUPAC Name3-benzyl-5-(2-hydroxyethyl)-4-methyl-1,3-thiazol-3-ium chloride
Molecular FormulaC13H16ClNOS
InChI KeyIWSVLBKHBJGMAA-UHFFFAOYSA-M
SMILES[Cl-].CC1=C(CCO)SC=[N+]1CC1=CC=CC=C1
さらに表示
仕様 スペックシート
スペックシート
Appearance (Color)White to cream
FormCrystals or powder or crystalline powder
Assay (Titration ex Chloride)≥97.5 to ≤102.5%
Identification (FTIR)Conforms
Water Content (Karl Fischer Titration)≤0.5%
3-Benzyl-5-(2-hydroxyethyl)-4-methylthiazolium chloride, aliphatic aldehydes undergo the acyloin condensation in the presence of this catalyst and mild base. It acts as a catalyst for the addition of aliphatic and heterocyclic aldehydes to α,β-unsaturated ketones, nitriles and esters.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
3-Benzyl-5-(2-hydroxyethyl)-4-methylthiazolium chloride, aliphatic aldehydes undergo the acyloin condensation in the presence of this catalyst and mild base. It acts as a catalyst for the addition of aliphatic and heterocyclic aldehydes to α,β-unsaturated ketones, nitriles and esters.
Solubility
Soluble in water.
Notes
Hygroscopic. Store away from water and oxidizing agents. Protect from humidity and water. Incompatible with strong oxidizing agents.
3-Benzyl-5-(2-hydroxyethyl)-4-methylthiazolium chloride, aliphatic aldehydes undergo the acyloin condensation in the presence of this catalyst and mild base. It acts as a catalyst for the addition of aliphatic and heterocyclic aldehydes to α,β-unsaturated ketones, nitriles and esters.
Solubility
Soluble in water.
Notes
Hygroscopic. Store away from water and oxidizing agents. Protect from humidity and water. Incompatible with strong oxidizing agents.
RUO – Research Use Only
General References:
- Aliphatic aldehydes undergo the acyloin condensation in the presence of this catalyst and mild base. For example (butyraldehyde to butyroin), see: Org. Synth. Coll., 7, 95 (1990).
- The same system is effective in catalyzing the addition of aldehydes to electron-deficient alkenes, including ɑß-unsaturated ketones, esters and nitriles (Stetter reaction), providing a non-toxic alternative to cyanide in these reactions; e.g. in the synthesis of dihydrojasmone: Org. Synth. Coll., 8, 620 (1993):
- For reviews, see: Angew. Chem. Int. Ed., 15, 639 (1976); Org. React., 40, 407 (1991). For a further example, see Phenyl vinyl sulfone, A14794.