Search
Thermo Scientific Chemicals
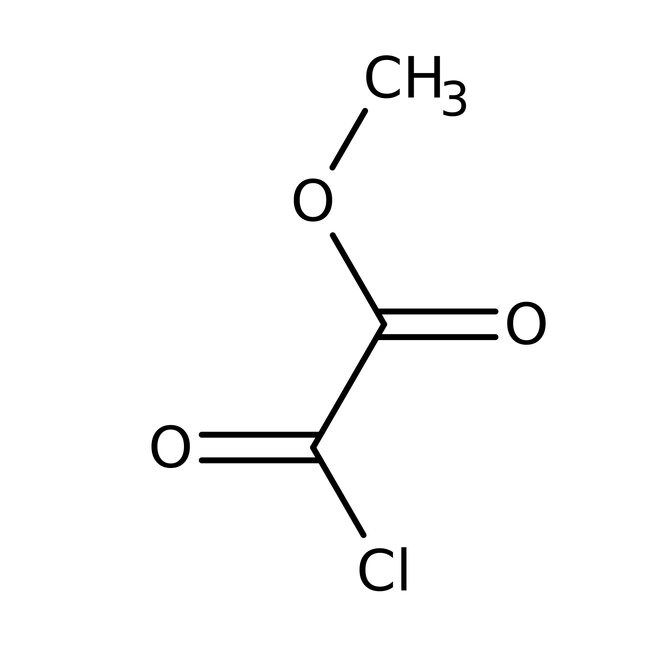
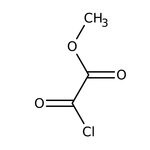
Methyl oxalyl chloride, 97%
CAS: 5781-53-3 | C3H3ClO3 | 122.504 g/mol
化学物質識別子
CAS5781-53-3
IUPAC Namemethyl oxalochloridate
Molecular FormulaC3H3ClO3
InChI KeyZXUQEPZWVQIOJE-UHFFFAOYSA-N
SMILESCOC(=O)C(Cl)=O
さらに表示
仕様 スペックシート
スペックシート
Appearance (Color)Clear colorless
Identification (FTIR)Conforms
FormLiquid
Assay (GC)≥96.0%
Refractive Index1.4155-1.4210 @ 20?C
Methyl oxalyl chloride is used as a synthetic reagent. It serves as a reagent in the synthesis of fused coumarins, substituted isoxazoles and heterocycles. Further, it is used in intramolecular Wittig reactions, and iron-mediated cleavage of C-C bonds.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Methyl oxalyl chloride is used as a synthetic reagent. It serves as a reagent in the synthesis of fused coumarins, substituted isoxazoles and heterocycles. Further, it is used in intramolecular Wittig reactions, and iron-mediated cleavage of C-C bonds.
Solubility
Miscible with water.
Notes
Moisture sensitive. Incompatible with water, oxidizing agents, alcohols and bases.
Methyl oxalyl chloride is used as a synthetic reagent. It serves as a reagent in the synthesis of fused coumarins, substituted isoxazoles and heterocycles. Further, it is used in intramolecular Wittig reactions, and iron-mediated cleavage of C-C bonds.
Solubility
Miscible with water.
Notes
Moisture sensitive. Incompatible with water, oxidizing agents, alcohols and bases.
RUO – Research Use Only
General References:
- Zhao, L.; Yang, H. J.; Cai, Z. Solubility comparison and partial molar volumes of 1, 2-hexanediol before and after end-group modification by methyl oxalyl chloride and ethyl oxalyl monochloride in supercritical CO2. J. Chem. Thermodyn. 2013, 57, 46-53.
- Fandrick, K. R.; Li, W.; Zhang, Y.; Tang, W.; Gao, J.; Rodriguez, S.; Patel, N. D.; Reeves, D. C.; Wu, J. P.; Sanyal, S.; Gonnella, N.; Qu, B.; Haddad, N.; Lorenz, J. C.; Sidhu, K.; Wang, J.; Ma, S.; Grinberg, N.; Lee, H.; Tsantrizos, Y.; Poupart, M. A.; Busacca, C. A.; Yee, N. K.; Lu, B. Z.; Senanayake, C. H. Concise and Practical Asymmetric Synthesis of a Challenging Atropisomeric HIV Integrase Inhibitor. Angew. Chem. 2015, 127 (24), 7250-7254.