Search
Thermo Scientific Chemicals
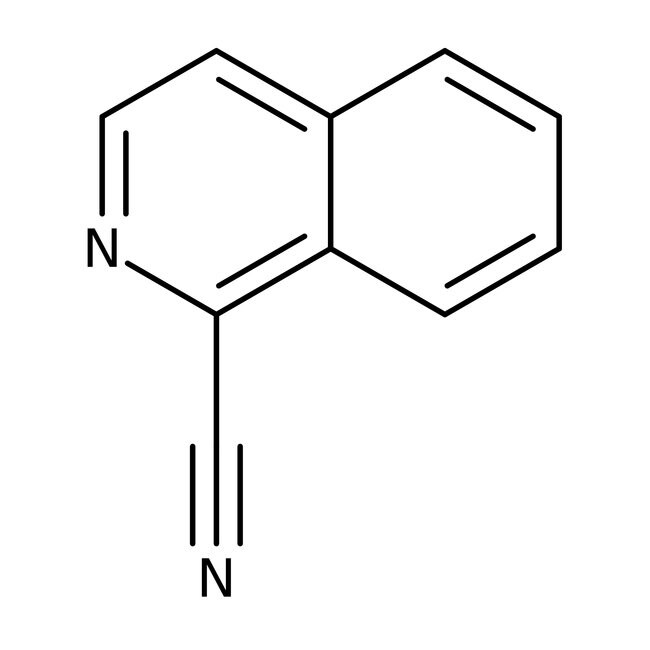
Isoquinoline-1-carbonitrile, 99%
CAS: 1198-30-7 | C10H6N2 | 154.172 g/mol
化学物質識別子
CAS1198-30-7
IUPAC Nameisoquinoline-1-carbonitrile
Molecular FormulaC10H6N2
InChI KeyHJHXYSBRTVFEDD-UHFFFAOYSA-N
SMILESN#CC1=C2C=CC=CC2=CC=N1
さらに表示
仕様 スペックシート
スペックシート
Appearance (Color)White to cream or pale yellow
Assay (GC)≥98.5%
FormCrystals or powder or crystalline powder or flakes
Identification (FTIR)Conforms
Isoquinoline-1-carbonitrile is used as starting reagent in the syntheses of imidazo[5,1-a]isoquinolines. It is also used as intermediate in organic syntheses.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Isoquinoline-1-carbonitrile is used as starting reagent in the syntheses of imidazo[5,1-a]isoquinolines. It is also used as intermediate in organic syntheses.
Solubility
Insoluble in water.
Notes
Store in cool place. Keep container tightly closed in a dry and well-ventilated place. Incompatible with oxidizing agents.
Isoquinoline-1-carbonitrile is used as starting reagent in the syntheses of imidazo[5,1-a]isoquinolines. It is also used as intermediate in organic syntheses.
Solubility
Insoluble in water.
Notes
Store in cool place. Keep container tightly closed in a dry and well-ventilated place. Incompatible with oxidizing agents.
RUO – Research Use Only
General References:
- Sharmistha Dutta Choudhury.; Samita Basu. Interaction of 4-nitroquinoline-1-oxide with indole derivatives and some related biomolecules: a study with magnetic field. Journal of Physical Chemistry. 2006, 110 (17), 8850-8855.
- Mickaël Pauvert.; Sylvain C. Colleta.; Marie-Jo Bertranda.; André Y. Guingant.; Michel Evain. Synthesis of chiral N-protected 1,2-dihydro-quinoline-2-carbonitrile and 1,2-dihydro-isoquinoline-1-carbonitrile via an asymmetric Reissert reaction. Tetrahedron Letters. 2005, 46 (17), 2983-2987.