Product References
Tenderization of Beef Semitendinosus Muscle by Pulsed Electric Field Treatment with a Direct Contact Chamber and Its Impact on Proteolysis and Physicochemical Properties.
Foods (Basel, Switzerland)
Jeong SH,Jung YM,Kim S,Kim JH,Yeo H,Lee DU
MA3-940 was used in Western Blotting to investigate the effects of pulse electric field (PEF) treatment on the tenderization of beef semitendinosus muscle.
Tue Jan 17 00:00:00 EST 2023
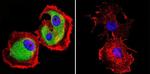
Endosomal phosphatidylinositol 3-phosphate controls synaptic vesicle cycling and neurotransmission.
The EMBO journal
Liu GT,Kochlamazashvili G,Puchkov D,Müller R,Schultz C,Mackintosh AI,Vollweiter D,Haucke V,Soykan T
Published figure using Mu-Calpain monoclonal antibody (Product # MA3-940) in Immunocytochemistry
Mon May 02 00:00:00 EDT 2022
Elucidating the involvement of apoptosis in postmortem proteolysis in porcine muscles from two production cycles using metabolomics approach.
Scientific reports
Ma D,Suh DH,Zhang J,Chao Y,Duttlinger AW,Johnson JS,Lee CH,Kim YHB
MA3-940 was used in Western Blot to evaluate apoptotic and proteolytic characteristics and metabolome changes of porcine muscles in a muscle-meat conversion prospective.
Wed Feb 10 00:00:00 EST 2021
Elucidating the involvement of apoptosis in postmortem proteolysis in porcine muscles from two production cycles using metabolomics approach.
Scientific reports
Ma D,Suh DH,Zhang J,Chao Y,Duttlinger AW,Johnson JS,Lee CH,Kim YHB
MA3-940 was used in Western Blot to evaluate apoptotic and proteolytic characteristics and metabolome changes of porcine muscles in a muscle-meat conversion prospective.
Wed Feb 10 00:00:00 EST 2021
Impact of Feeding Cover Crop Forage Containing Brassicas to Steers during Backgrounding on Palatability Attributes of Beef Strip Steaks.
Foods (Basel, Switzerland)
Bakker C,Hite L,Wright C,Smart A,Dinh T,Blair A,Underwood K,Grubbs JK
MA3-940 was used in Western Blot to investigate the impact of feeding a brassica cover crop mixture during backgrounding on beef palatability.
Mon May 31 00:00:00 EDT 2021
Impact of Feeding Cover Crop Forage Containing Brassicas to Steers during Backgrounding on Palatability Attributes of Beef Strip Steaks.
Foods (Basel, Switzerland)
Bakker C,Hite L,Wright C,Smart A,Dinh T,Blair A,Underwood K,Grubbs JK
MA3-940 was used in Western Blot to investigate the impact of feeding a brassica cover crop mixture during backgrounding on beef palatability.
Mon May 31 00:00:00 EDT 2021
Cardiomyocyte specific deletion of p53 decreases cell injury during ischemia-reperfusion: Role of Mitochondria.
Free radical biology & medicine
Chen Q,Thompson J,Hu Y,Lesnefsky EJ
MA3-940 was used in Western Blot to conclude that deletion of p53 decreases cardiac injury by protecting mitochondria through attenuation of oxidative stress and calpain activation during ischemia-reperfusion.
Thu Oct 01 00:00:00 EDT 2020
Inhibition of the ubiquitous calpains protects complex I activity and enables improved mitophagy in the heart following ischemia-reperfusion.
American journal of physiology. Cell physiology
Chen Q,Thompson J,Hu Y,Dean J,Lesnefsky EJ
MA3-940 was used in Western Blotting to show that activation of both cytosolic and mitochondrial calpains impairs mitochondria during cardiac ISC-REP. Mitochondria-localised calpains impair complex I via cleavage for a key subunit.
Fri Nov 01 00:00:00 EDT 2019
Effect of fast freezing then thaw-aging on meat quality attributes of lamb M. longissimus lumborum.
Animal science journal = Nihon chikusan Gakkaiho
Balan P,Kim YHB,Stuart AD,Kemp R,Staincliffe M,Craigie C,Farouk MM
MA3-940 was used in Western Blotting to demonstrate that fast freezing then thaw-aging improves water-holding capacity and tenderness by minimizing extracellular ice crystal formation, reducing purge and drip losses, and improving proteolysis in thawed lamb.
Thu Aug 01 00:00:00 EDT 2019
Influence of feed efficiency classification and growing and finishing diet type on meat tenderness attributes of beef steers.
Journal of animal science
Blank CP,Russell J,Lonergan SM,Hansen SL
MA3-940 was used in Western Blotting to indicate that high-fiber diets may negatively impact meat tenderness, and further work is needed to clarify the influence of diet type on parameters of meat tenderness.
Sat Jul 01 00:00:00 EDT 2017
Forage legumes rich in condensed tannins may increase n-3 fatty acid levels and sensory quality of lamb meat.
Journal of the science of food and agriculture
Girard M,Dohme-Meier F,Silacci P,Ampuero Kragten S,Kreuzer M,Bee G
MA3-940 was used in western blot to study the effects of tannins on meat quality
Fri Apr 01 00:00:00 EDT 2016
Differences in calpain system, desmin degradation and water holding capacity between commercial Meishan and Duroc × Landrace × Yorkshire crossbred pork.
Animal science journal = Nihon chikusan Gakkaiho
Wang J,Yan XL,Liu R,Fu QQ,Zhou GH,Zhang WG
MA3-940 was used in western blot to investigate the differences in calpain system, desmin degradation, pH values and water holding capacity between muscles of commercial Meishan and Duroc? x ?Landrace? x ?Yorkshire crossbred pigs
Fri Jan 01 00:00:00 EST 2016
Effect of early postmortem enhancement of calcium lactate/phosphate on quality attributes of beef round muscles under different packaging systems.
Meat science
Cruzen SM,Kim YH,Lonergan SM,Grubbs JK,Fritchen AN,Huff-Lonergan E
MA3-940 was used in western blot to determine the effect of calcium lactate/phosphate enhancement on the quality of beef round cuts in high-oxygen modified atmosphere
Sun Mar 01 00:00:00 EST 2015
The development of meat tenderness is likely to be compartmentalised by ultimate pH.
Meat science
Lomiwes D,Farouk MM,Wu G,Young OA
MA3-940 was used in western blot to study the effects of ultimate pH on the development of meat tenderness in Bulls
Wed Jan 01 00:00:00 EST 2014
Postmortem proteolysis in three muscles from growing and mature beef cattle.
Meat science
Cruzen SM,Paulino PV,Lonergan SM,Huff-Lonergan E
MA3-940 was used in western blot to study proteolysis and calpain/calpastatin in three postmortem muscles from young and mature beef cattle and the implications for meat tenderness
Sat Feb 01 00:00:00 EST 2014
The protection of bovine skeletal myofibrils from proteolytic damage post mortem by small heat shock proteins.
Meat science
Lomiwes D,Hurst SM,Dobbie P,Frost DA,Hurst RD,Young OA,Farouk MM
MA3-940 was used in western blot to study the role of small heat shock proteins in protecting bovine skeletal myofibrils from post-mortem proteolytic degradation
Fri Aug 01 00:00:00 EDT 2014
Influence of supplemental vitamin C on postmortem protein degradation and fatty acid profiles of the longissimus thoracis of steers fed varying concentrations of dietary sulfur.
Meat science
Pogge DJ,Lonergan SM,Hansen SL
MA3-940 was used in western blot to study postmortem muscle proteolysis and fatty acid profiles in beef cattle receiving different amounts of dietary sulfur and the effects of supplemental vitamin C
Sat Feb 01 00:00:00 EST 2014
Influence of supplementing vitamin C to yearling steers fed a high sulfur diet during the finishing period on meat color, tenderness and protein degradation, and fatty acid profile of the longissimus muscle.
Meat science
Pogge DJ,Lonergan SM,Hansen SL
MA3-940 was used in western blot to study the effect of finishing period vitamin C supplementation on various measures of meat quality in yearling steers fed a diet high in sulphur
Fri Aug 01 00:00:00 EDT 2014
Effect of nitric oxide on μ-calpain activation, protein proteolysis, and protein oxidation of pork during post-mortem aging.
Journal of agricultural and food chemistry
Li YP,Liu R,Zhang WG,Fu QQ,Liu N,Zhou GH
MA3-940 was used in western blot to investigate the effect of nitric oxide (NO) on calpain activation, protein proteolysis, and oxidation in porcine muscles
Wed Jun 25 00:00:00 EDT 2014
Comparison of protein degradation, protein oxidation, and μ-calpain activation between pale, soft, and exudative and red, firm, and nonexudative pork during postmortem aging.
Journal of animal science
Yin Y,Zhang WG,Zhou GH,Guo B
MA3-940 was used in western blot to investigate the differences in protein modifications between pale, soft, and exudative and red, firm, and nonexudative pork during postmortem aging
Fri Aug 01 00:00:00 EDT 2014
Pre rigor processing, ageing and freezing on tenderness and colour stability of lamb loins.
Meat science
Kim YH,Luc G,Rosenvold K
MA3-940 was used in western blot to study the effect of different aging and freezing protocols on lamb loin tenderness and color stability
Tue Oct 01 00:00:00 EDT 2013
Evaluation of feedlot cattle working chute behavior relative to temperament, tenderness, and postmortem proteolysis.
Meat science
Magolski JD,Berg EP,Hall NL,Anderson VL,Keller WL,Jeske TM,Carlin KR
MA3-940 was used in western blot to study whether calpain activation and post-mortem proteolysis play any role in the relationship between cattle feed chute behaviour and meat tenderness
Sun Sep 01 00:00:00 EDT 2013
Small heat shock proteins and toughness in intermediate pHu beef.
Meat science
Lomiwes D,Farouk MM,Frost DA,Dobbie PM,Young OA
MA3-940 was used in western blot to study the role of small heat shock proteins in toughness of beef with an intermediate ultimate pH
Fri Nov 01 00:00:00 EDT 2013
Distinct roles for μ-calpain and m-calpain in synaptic NMDAR-mediated neuroprotection and extrasynaptic NMDAR-mediated neurodegeneration.
The Journal of neuroscience : the official journal of the Society for Neuroscience
Wang Y,Briz V,Chishti A,Bi X,Baudry M
MA3-940 was used in western blot to study the distinct roles of different calpain isoforms in synaptic neuroprotection and extrasynaptic neurodegeneration mediated by the NMDA receptor
Wed Nov 27 00:00:00 EST 2013
Evidence of decreased muscle protein turnover in gilts selected for low residual feed intake.
Journal of animal science
Cruzen SM,Harris AJ,Hollinger K,Punt RM,Grubbs JK,Selsby JT,Dekkers JC,Gabler NK,Lonergan SM,Huff-Lonergan E
MA3-940 was used in western blot to study the rate of protein turnover in finisher pigs receiving low residual feed intake
Thu Aug 01 00:00:00 EDT 2013
Profile of biochemical traits influencing tenderness of muscles from the beef round.
Meat science
Anderson MJ,Lonergan SM,Fedler CA,Prusa KJ,Binning JM,Huff-Lonergan E
MA3-940 was used in western blot to study the biochemical factors governing commercially important beef cut tenderness
Sun Jul 01 00:00:00 EDT 2012
High pre rigor temperature limits the ageing potential of beef that is not completely overcome by electrical stimulation and muscle restraining.
Meat science
Kim YH,Stuart A,Nygaard G,Rosenvold K
MA3-940 was used in western blot to study the effects on beef quality of electrical input, wrapping, pre rigor temperature and different post rigor chilling rates
Tue May 01 00:00:00 EDT 2012
Involvement of calpain 2 in ionomycin-induced cell death in cultured mouse lens epithelial cells.
Current eye research
Nakajima T,Shearer TR,Azuma M
MA3-940 was used in western blot to investigate the role of calpain 2 in the ionomycin-induced cell death in mouse lens epithelial cells
Sat Oct 01 00:00:00 EDT 2011
Joubert syndrome Arl13b functions at ciliary membranes and stabilizes protein transport in Caenorhabditis elegans.
The Journal of cell biology
Cevik S,Hori Y,Kaplan OI,Kida K,Toivenon T,Foley-Fisher C,Cottell D,Katada T,Kontani K,Blacque OE
MA3-940 was used in western blot to investigate the role of Arl13b in ciliary protein transport in C. elegans
Mon Mar 22 00:00:00 EDT 2010
High-oxygen modified atmosphere packaging system induces lipid and myoglobin oxidation and protein polymerization.
Meat science
Kim YH,Huff-Lonergan E,Sebranek JG,Lonergan SM
MA3-940 was used in western blot to investigate the effect of the high-oxygen modified atmosphere packaging system on oxidation and polymerization
Sun Aug 01 00:00:00 EDT 2010
Protein denaturing conditions in beef deep semimembranosus muscle results in limited μ-calpain activation and protein degradation.
Meat science
Kim YH,Lonergan SM,Huff-Lonergan E
MA3-940 was used in western blot to investigate the effect of protein denaturation on u-calpain activation
Mon Nov 01 00:00:00 EDT 2010
Protease activity in post-mortem red swamp crayfish (Procambarus clarkii) muscle stored in modified atmosphere packaging.
Journal of agricultural and food chemistry
Chen G,Guttmann RP,Xiong YL,Webster CD,Romaire RP
MA3-940 was used in western blot to study the involvement of protease activity in storage of red swamp crayfish
Wed Sep 24 00:00:00 EDT 2008
Role of mu-calpain in human decidua for recurrent miscarriage.
American journal of reproductive immunology (New York, N.Y. : 1989)
Kumagai K,Ozaki Y,Nakanishi T,Inomata M,Furuno T,Nakanishi M,Ogasawara MS
MA3-940 was used in western blot to study the role of mu-calpain in decidua from patients with recurrent miscarriage
Tue Apr 01 00:00:00 EDT 2008
Rate and extent of pH decline affect proteolysis of cytoskeletal proteins and water-holding capacity in pork.
Meat science
Bee G,Anderson AL,Lonergan SM,Huff-Lonergan E
MA3-940 was used in western blot to study the effect of pH changes on breakdown of porcine cytoskeletal proteins
Fri Jun 01 00:00:00 EDT 2007
Effect of postmortem storage on activity of mu- and m-calpain in five bovine muscles.
Journal of animal science
Camou JP,Marchello JA,Thompson VF,Mares SW,Goll DE
MA3-940 was used in western blot to evaluate the effect of the postmortem storage time and temperature on calpain activity in bovine muscles
Mon Oct 01 00:00:00 EDT 2007
Isolation and characterization of mu-calpain, m-calpain, and calpastatin from postmortem muscle. I. Initial steps.
Journal of animal science
Camou JP,Mares SW,Marchello JA,Vazquez R,Taylor M,Thompson VF,Goll DE
MA3-940 was used in western blot to isolate and characterize calpain isoforms and calpastatin
Sat Dec 01 00:00:00 EST 2007
Purification and characterization of calpain and calpastatin from rainbow trout, Oncorhynchus mykiss.
Comparative biochemistry and physiology. Part B, Biochemistry & molecular biology
Saito M,Li H,Thompson VF,Kunisaki N,Goll DE
MA3-940 was used in western blot to purify and characterize the calpain and calpastatin from rainbow trout
Sun Apr 01 00:00:00 EDT 2007
Contribution of postmortem changes of integrin, desmin and μ-calpain to variation in water holding capacity of pork.
Meat science
Zhang WG,Lonergan SM,Gardner MA,Huff-Lonergan E
MA3-940 was used in western blot to investigate the postmortem changes of integrin, desmin, and calpain in pork
Wed Nov 01 00:00:00 EST 2006
Effects of 25-hydroxyvitamin D3 and manipulated dietary cation-anion difference on the tenderness of beef from cull native Korean cows.
Journal of animal science
Cho YM,Choi H,Hwang IH,Kim YK,Myung KH
MA3-940 was used in western blot to study the effect of 25-hydroxyvitamin D3 and manipulated dietary cation-anion difference on beef quality
Thu Jun 01 00:00:00 EDT 2006
Calpain may contribute to hereditary cataract formation in sheep.
Investigative ophthalmology & visual science
Robertson LJ,Morton JD,Yamaguchi M,Bickerstaffe R,Shearer TR,Azuma M
MA3-940 was used in western blot to investigate the role of calpain during hereditary cataract formation in sheep
Thu Dec 01 00:00:00 EST 2005
Effect of pH and ionic strength on mu- and m-calpain inhibition by calpastatin.
Journal of animal science
Maddock KR,Huff-Lonergan E,Rowe LJ,Lonergan SM
MA3-940 was used in western blot to study the effect of pH and ionic strength on mu- and m-calpain activity and the ability of calpastatin to inhibit the activity of mu- or m-calpain
Wed Jun 01 00:00:00 EDT 2005
Oxidative environments decrease tenderization of beef steaks through inactivation of mu-calpain.
Journal of animal science
Rowe LJ,Maddock KR,Lonergan SM,Huff-Lonergan E
MA3-940 was used in western blot to investigate the role of inactivation of mu-calpain for tenderization of beef steaks during oxidative environments
Mon Nov 01 00:00:00 EST 2004
Early postmortem biochemical factors influence tenderness and water-holding capacity of three porcine muscles.
Journal of animal science
Melody JL,Lonergan SM,Rowe LJ,Huiatt TW,Mayes MS,Huff-Lonergan E
MA3-940 was used in immunohistochemistry to investigate the effect of early postmortem biochemical factors on tenderness and water-holding capacity of three porcine muscles
Thu Apr 01 00:00:00 EST 2004
The calpain system in human placenta.
Life sciences
Thompson VF,Saldaña S,Cong J,Luedke DM,Goll DE
MA3-940 was used in western blot to study the calpains present in human placenta
Sun Apr 21 00:00:00 EDT 2002
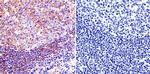
Fiber type-specific expression of major proteolytic systems in fast- to slow-transforming rabbit muscle.
American journal of physiology. Cell physiology
Sultan KR,Dittrich BT,Leisner E,Paul N,Pette D
MA3-940 was used in immunohistochemistry to study the importance of two major proteolytic systems in tranforming rabbit and rat muscles
Thu Feb 01 00:00:00 EST 2001
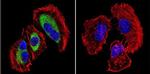
The endoplasmic reticulum chaperone glycoprotein GRP94 with Ca(2+)-binding and antiapoptotic properties is a novel proteolytic target of calpain during etoposide-induced apoptosis.
The Journal of biological chemistry
Reddy RK,Lu J,Lee AS
MA3-940 was used in immunocytochemistry to show that a calcium-binding ER protein with protective functions against calcium-induced apoptosis is a substrate for a calcium-activated protease
Fri Oct 01 00:00:00 EDT 1999
Changes in the calpains and calpastatin during postmortem storage of bovine muscle.
Journal of animal science
Boehm ML,Kendall TL,Thompson VF,Goll DE
MA3-940 was used in western blot to detect the changes of micro-calpain, m-calpain, and calpastatin in bovine muscle during postmortem storage
Tue Sep 01 00:00:00 EDT 1998
Effect of monoclonal antibodies specific for the 28-kDa subunit on catalytic properties of the calpains.
The Journal of biological chemistry
Cong J,Thompson VF,Goll DE
MA3-940 was used in western blot to study the effect of specific calpain antibodies for the regulation of calpain function
Sun Dec 05 00:00:00 EST 1993
A comparison of the intracellular distribution of mu-calpain, m-calpain, and calpastatin in proliferating human A431 cells.
Experimental cell research
Lane RD,Allan DM,Mellgren RL
MA3-940 was used in immunocytochemistry to compare the cellular localization of calpains and calpastatin in human A431 cells
Sun Nov 01 00:00:00 EST 1992










![]() cysteine protease
cysteine protease



