Search
Thermo Scientific Chemicals
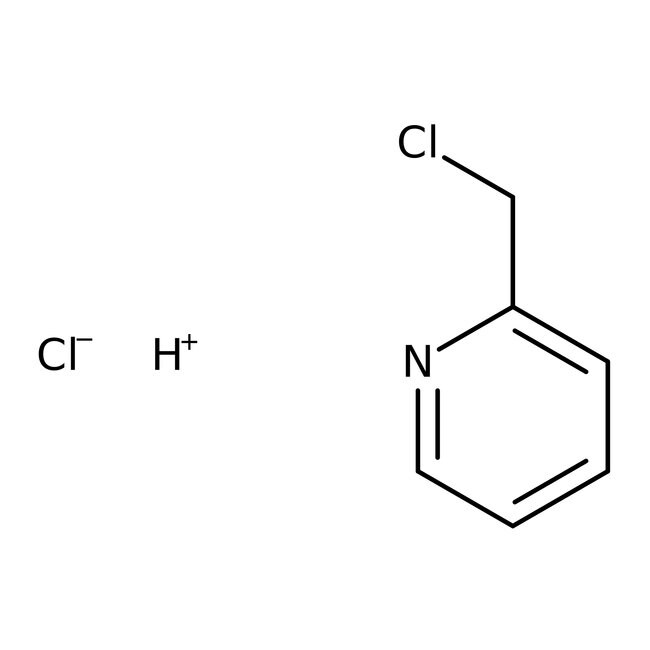
2-(Chloromethyl)pyridine hydrochloride, 98%
CAS: 6959-47-3 | C6H7Cl2N | 164.029 g/mol
Catalog number A10226.22
also known as A10226-22
Price (USD)
304.65
Online Exclusive
338.00Save 33.35 (10%)
Each
Quantity:
100 g
Price (USD)
304.65
Online Exclusive
338.00Save 33.35 (10%)
Each
Chemical Identifiers
CAS6959-47-3
IUPAC Namehydrogen 2-(chloromethyl)pyridine chloride
Molecular FormulaC6H7Cl2N
InChI KeyJPMRGPPMXHGKRO-UHFFFAOYSA-N
SMILES[H+].[Cl-].ClCC1=CC=CC=N1
View more
Specifications Specification Sheet
Specification Sheet
Assay (Titration ex Chloride)≥97.5 to ≤102.5%
Water Content (Karl Fischer Titration)≤2.5%
Solution Test0.5g in 20ml Ethanol to be transparent or almost transparent
Appearance (Color)White to yellow or pale brown
FormCrystals or powder or crystalline powder
2-(Chloromethyl)pyridine hydrochloride was used as reagent in base catalyzed alkylation of p-tert-butylcalix[6]arene and p-tert-butylcalix[5]arene in DMF. It was used in the synthesis of Gd3+ diethylenetriaminepentaacetic acid bisamide complex, a Zn2+-sensitive magnetic resonance imaging contrast agent.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
2-(Chloromethyl)pyridine hydrochloride was used as reagent in base catalyzed alkylation of p-tert-butylcalix[6]arene and p-tert-butylcalix[5]arene in DMF. It was used in the synthesis of Gd3+ diethylenetriaminepentaacetic acid bisamide complex, a Zn2+-sensitive magnetic resonance imaging contrast agent.
Solubility
Soluble in water, acetone (10-50mg/ml), 95%ethanol (100mg/ml).
Notes
Hygroscopic store in inert gas Store away from strong oxidizing agents. keep container tightly closed. Store in cool, dry conditions in well sealed containers.
2-(Chloromethyl)pyridine hydrochloride was used as reagent in base catalyzed alkylation of p-tert-butylcalix[6]arene and p-tert-butylcalix[5]arene in DMF. It was used in the synthesis of Gd3+ diethylenetriaminepentaacetic acid bisamide complex, a Zn2+-sensitive magnetic resonance imaging contrast agent.
Solubility
Soluble in water, acetone (10-50mg/ml), 95%ethanol (100mg/ml).
Notes
Hygroscopic store in inert gas Store away from strong oxidizing agents. keep container tightly closed. Store in cool, dry conditions in well sealed containers.
RUO – Research Use Only
General References:
- Neri P and Pappalardo S. Functionalization of p-tert-butylcalix [6] arene by alkylation with 2-(chloromethyl) pyridine hydrochloride. J. Org. Chem. 1993, 58,(5), 1048-53.
- Pappalardo S and Ferguson G. Functionalization of p-tert-Butylcalix [5] arene by Alkylation with 2-(Chloromethyl) pyridine Hydrochloride. J. Org. Chem. 1996, 61,(7),2407-12.
- Hanaoka K, et al. Selective sensing of zinc ions with a novel magnetic resonance imaging contrast agent. J. Chem. Soc. Perkin Trans. 2001, 9,1840-43.
- The free base, generated from the hydrochloride with aqueous OH-, can be metallated (LDA, -78°) at the methylene group. Subsequent reaction with a ketone provides a route to otherwise inaccessible pyridyloxiranes: Tetrahedron Lett., 35, 3175 (1994). Similarly, reaction of the lithio derivative with an imine gives a pyridylaziridine, generally in good yield: J. Org. Chem., 60, 2279 (1995).
- The free base has also been used for the protection of OH groups as 2-picolyl derivatives, relatively stable to acid (HF, TFA) but selectively cleaved by electrolytic reduction: Acta Chem. Scand. B, B37, 475 (1983); J. Chem. Res. (Synop.), 22 (1977).
- For a study of the base-catalyzed alkylation of p-t-butylcalix[5]arene, see: J. Org. Chem., 61, 2407 (1996).