Search
Thermo Scientific Chemicals
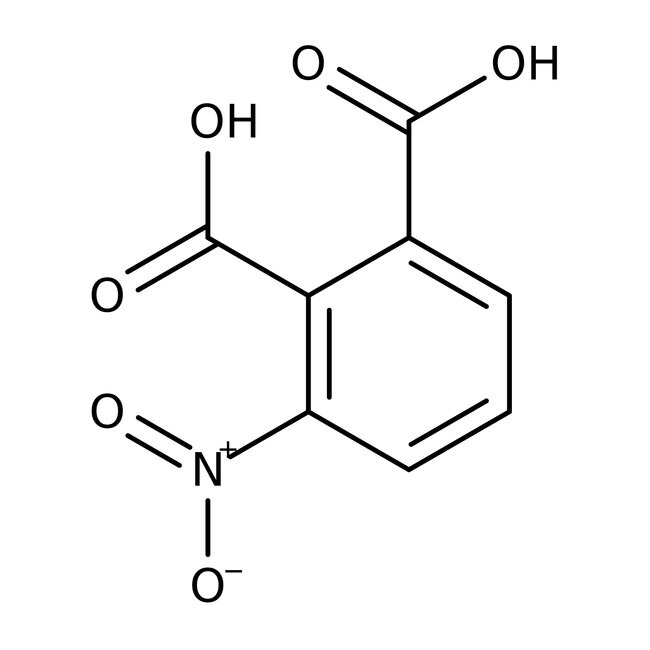
3-Nitrophthalic acid, 95%
CAS: 603-11-2 | C8H5NO6 | 211.129 g/mol
Catalog number A14281.22
also known as A14281-22
Price (USD)
51.65
Special offer
Online exclusive
Ends: 30-Jun-2026
60.90Save 9.25 (15%)
Each
Quantity:
100 g
Price (USD)
51.65
Special offer
Online exclusive
Ends: 30-Jun-2026
60.90Save 9.25 (15%)
Each
Chemical Identifiers
CAS603-11-2
IUPAC Name3-nitrobenzene-1,2-dicarboxylic acid
Molecular FormulaC8H5NO6
InChI KeyKFIRODWJCYBBHY-UHFFFAOYSA-N
SMILESOC(=O)C1=CC=CC(=C1C(O)=O)[N+]([O-])=O
View more
Specifications Specification Sheet
Specification Sheet
Appearance (Color)White to pale cream to pale yellow
FormCrystals or powder or crystalline powder
Assay (HPLC)≥94.0%
Assay (Aqueous acid-base Titration)≥94.0 to ≤106.0%
3-Nitrophthalic acid can be used as starting reagent in the synthesis of modified 2-iodoxybenzoic acid derivatives. It may be used in the synthesis of brucinium 2-carboxy-6-nitrophthalate dihydrate (systematic name: 2,3-dimethoxy-10-oxostrychnidinium 2-carboxy-6-nitrophthalate dihydrate), via heating with brucine. Clyatt E
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
3-Nitrophthalic acid can be used as starting reagent in the synthesis of modified 2-iodoxybenzoic acid derivatives. It may be used in the synthesis of brucinium 2-carboxy-6-nitrophthalate dihydrate (systematic name: 2,3-dimethoxy-10-oxostrychnidinium 2-carboxy-6-nitrophthalate dihydrate), via heating with brucine. Clyatt E
Solubility
Solubility in methanol is almost transparent. Soluble in 5% water.
Notes
Store away from oxidizing agents. Keep the container tightly closed and place it in a cool, dry and well ventilated condition.
3-Nitrophthalic acid can be used as starting reagent in the synthesis of modified 2-iodoxybenzoic acid derivatives. It may be used in the synthesis of brucinium 2-carboxy-6-nitrophthalate dihydrate (systematic name: 2,3-dimethoxy-10-oxostrychnidinium 2-carboxy-6-nitrophthalate dihydrate), via heating with brucine. Clyatt E
Solubility
Solubility in methanol is almost transparent. Soluble in 5% water.
Notes
Store away from oxidizing agents. Keep the container tightly closed and place it in a cool, dry and well ventilated condition.
RUO – Research Use Only
General References:
- Tohma H and Kita Y. Hypervalent iodine reagents for the oxidation of alcohols and their application to complex molecule synthesis.Adv. Synth. Catal.,2004,346(2-3), 111-124.
- Smith G, et al. Brucinium 2-carboxy-6-nitrophthalate dihydrate: the 1: 1 proton-transfer compound of brucine with 3-nitrophthalic acid.Acta Crystallogr. Sect. E Struct. Rep. Online.,2005,61(7), 2008-11.
- Drakeford, et. al. Fate of isoxaben in a containerized plant rhizosphere system.Chemosphere.,2003,50(9), 1243-1247.