Search
Thermo Scientific Chemicals
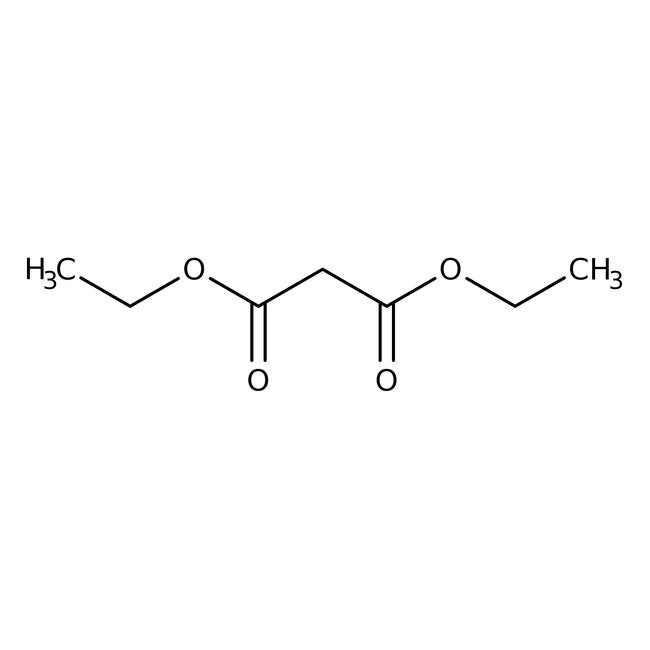
Diethyl malonate, 99%
CAS: 105-53-3 | C7H12O4 | 160.169 g/mol
Catalog number A15468.36
also known as A15468-36
Price (USD)
39.65
Online Exclusive
44.00Save 4.35 (10%)
Each
Quantity:
500 g
Price (USD)
39.65
Online Exclusive
44.00Save 4.35 (10%)
Each
Chemical Identifiers
CAS105-53-3
IUPAC Name1,3-diethyl propanedioate
Molecular FormulaC7H12O4
InChI KeyIYXGSMUGOJNHAZ-UHFFFAOYSA-N
SMILESCCOC(=O)CC(=O)OCC
View more
Specifications Specification Sheet
Specification Sheet
Assay (GC)≥98.5%
Water Content (Karl Fischer Titration)≤0.5%
Appearance (Color)Clear colorless
Refractive Index1.4125-1.4155 @ 20?C
Identification (FTIR)Conforms
View more
Diethyl malonate is used in organic synthesis for the preparation of alpha-aryl malonates, mono-substituted and di-substituted acetic acid, barbiturates and artificial flavorings. It is also involved in the synthesis of pharmaceuticals like chloroquine, butazolidin and barbital. It acts as intermediate in the synthesis of vitamin B1, vitamin B6, non-steroidal anti-inflammatory agents agrochemicals and perfumes. In Knoevenagel condensation reaction, it reacts with benzaldehyde to get diethyl benzylidenemalonate.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Diethyl malonate is used in organic synthesis for the preparation of alpha-aryl malonates, mono-substituted and di-substituted acetic acid, barbiturates and artificial flavorings. It is also involved in the synthesis of pharmaceuticals like chloroquine, butazolidin and barbital. It acts as intermediate in the synthesis of vitamin B1, vitamin B6, non-steroidal anti-inflammatory agents agrochemicals and perfumes. In Knoevenagel condensation reaction, it reacts with benzaldehyde to get diethyl benzylidenemalonate.
Solubility
Miscible with ethyl alcohol, ether, chloroform and benzene. Slightly miscible with water.
Notes
Incompatible with acids, bases, oxidizing agents and reducing agents.
Diethyl malonate is used in organic synthesis for the preparation of alpha-aryl malonates, mono-substituted and di-substituted acetic acid, barbiturates and artificial flavorings. It is also involved in the synthesis of pharmaceuticals like chloroquine, butazolidin and barbital. It acts as intermediate in the synthesis of vitamin B1, vitamin B6, non-steroidal anti-inflammatory agents agrochemicals and perfumes. In Knoevenagel condensation reaction, it reacts with benzaldehyde to get diethyl benzylidenemalonate.
Solubility
Miscible with ethyl alcohol, ether, chloroform and benzene. Slightly miscible with water.
Notes
Incompatible with acids, bases, oxidizing agents and reducing agents.
RUO – Research Use Only
General References:
- For phase-transfer monoalkylation of the active methylene group see, e.g. (18-crown-6): Synthesis, 37 (1977); and (BTEAC): Org. Prep. Proced. Int., 26, 469 (1994). Solid-liquid phase transfer alkylation has been promoted by microwave irradiation: Synth. Commun., 25,1761 (1995). Dialkylation can also occur in the presence of BTEAC: Synthesis, 54 (1985); Org. Synth. Coll., 7, 411 (1990), to give cyclopropane derivatives. Stereoselective alkylation has been achieved under particularly mild conditions with secondary alkyl mesylates in the presence of Cesium fluoride, 12885 in DMF; complete inversion occurs at the secondary alkyl carbon: J. Org. Chem., 60, 2627 (1995); see also: Synlett, 499 (1998).
- Substituents can also be introduced by Michael addition; see e.g.: Org. Synth. Coll., 8, 467 (1993).
- Arylation occurs with aryl bromides in the presence of CuI: Gazz. Chim. Ital., 122, 511 (1992).
- C-Acylation of malonic esters is normally carried out by reaction of the Mg derivative with an acid chloride. A convenient method using a combination of MgCl2 and triethylamine was introduced by Rathke: J. Org. Chem., 50, 2622 (1985); see also Triethyl amine, A12646 for discussion of this system:
- A number of procedures have been devised for the direct conversion of malonic esters to acetic esters (decarboalkoxylation), in a single step of which heating in wet DMSO is perhaps the simplest: J. Org. Chem., 43, 138 (1978). See also 1,4-Diazabicyclo[2.2.2]octane, A14003.
- Compare also Dimethyl malonate, A11007, Di-tert-butyl malonate, A12774, Dibenzyl malonate, A10844, and Isopropyl idene malonate, A15603.