Search
Thermo Scientific Chemicals
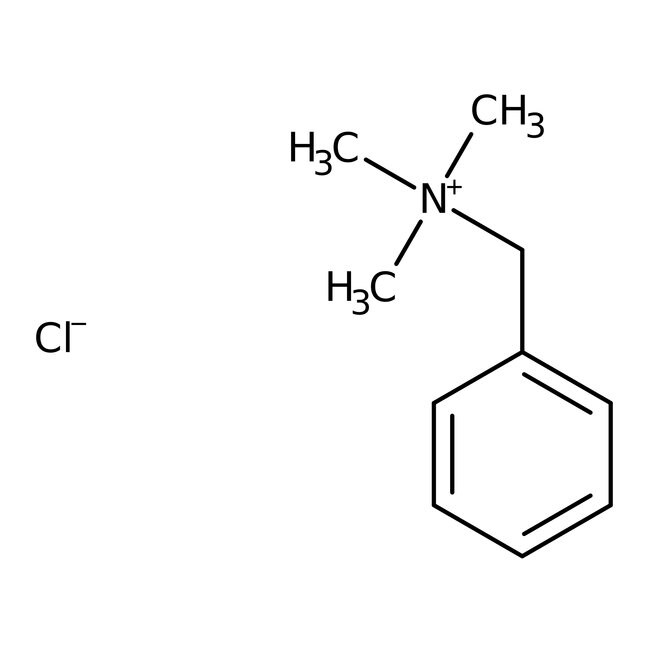
Benzyltrimethylammonium chloride, 97%
CAS: 56-93-9 | C10H16ClN | 185.70 g/mol
Catalog number A16759.22
also known as A16759-22
Price (USD)
29.65
Online Exclusive
32.60Save 2.95 (9%)
Each
Quantity:
100 g
Price (USD)
29.65
Online Exclusive
32.60Save 2.95 (9%)
Each
Chemical Identifiers
CAS56-93-9
IUPAC Namebenzyltrimethylazanium chloride
Molecular FormulaC10H16ClN
InChI KeyKXHPPCXNWTUNSB-UHFFFAOYSA-M
SMILES[Cl-].C[N+](C)(C)CC1=CC=CC=C1
View more
Specifications Specification Sheet
Specification Sheet
Appearance (Color)White
FormCrystals or powder or crystalline powder or lumps
Assay (Titration ex Chloride)≥96.0 to ≤104.0%
Water Content (Karl Fischer Titration)≤1.5%
Benzyltrimethylammonium chloride are Ccommercially important catalyst. Used in antistatic agent, detergent Sanitisers, softner for textiles and paper products, phase transfer catalyst.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Benzyltrimethylammonium chloride are Ccommercially important catalyst. Used in antistatic agent, detergent Sanitisers, softner for textiles and paper products, phase transfer catalyst.
Solubility
Soluble in water, ethanol, and butanol and slightly soluble in butyl phthalate
Notes
Hygroscopic. Store in cool place. Keep container tightly closed in a dry and well-ventilated place. Store away from strong oxidizing agents, water, moisture.
Benzyltrimethylammonium chloride are Ccommercially important catalyst. Used in antistatic agent, detergent Sanitisers, softner for textiles and paper products, phase transfer catalyst.
Solubility
Soluble in water, ethanol, and butanol and slightly soluble in butyl phthalate
Notes
Hygroscopic. Store in cool place. Keep container tightly closed in a dry and well-ventilated place. Store away from strong oxidizing agents, water, moisture.
RUO – Research Use Only
General References:
- L Sangeetha Vedula et. al.Exploring biosynthetic diversity with trichodiene synthase. Archives of Biochemistry and Biophysics,. 2007 , 466(2),260-266 .
- Lena Edström et. al. Deformations of overloaded bands under pH-stable conditions in reversed phase chromatography. Journal of Chromatography A. 2011, 1218( (15), 1966-1973 .