Search
Thermo Scientific Chemicals
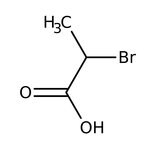
2-Bromopropionic acid, 98%
CAS: 598-72-1 | C3H5BrO2 | 152.98 g/mol
| Catalog Number | Quantity |
|---|---|
| A17569.30 also known as A17569-30 | 250 g |
Catalog number A17569.30
also known as A17569-30
Price (USD)
70.00
Each
Quantity:
250 g
Price (USD)
70.00
Each
Chemical Identifiers
CAS598-72-1
IUPAC Name2-bromopropanoic acid
Molecular FormulaC3H5BrO2
InChI KeyMONMFXREYOKQTI-UHFFFAOYNA-N
SMILESCC(Br)C(O)=O
View more
Specifications Specification Sheet
Specification Sheet
Appearance (Color)White or translucent to pale yellow
Assay (Aqueous acid-base Titration)≥97.5 to ≤102.5%
FormLow-melting fused solid (clear liquid as melt)
Identification (FTIR)Conforms
Assay (GC)≥97.5%
View more
It is employed as pharmaceutical and agrochemical intermediate. It is used as an alkylation agent for mercaptans and other sulfur containing compounds.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
It is employed as pharmaceutical and agrochemical intermediate. It is used as an alkylation agent for mercaptans and other sulfur containing compounds.
Solubility
Soluble in water.
Notes
Store away from oxidizing agents and strong bases. Keep the container tightly closed in a cool, dry place.
It is employed as pharmaceutical and agrochemical intermediate. It is used as an alkylation agent for mercaptans and other sulfur containing compounds.
Solubility
Soluble in water.
Notes
Store away from oxidizing agents and strong bases. Keep the container tightly closed in a cool, dry place.
RUO – Research Use Only
General References:
- H Xiao.; J Liu.; Z Li. Catalysis of the hydrolysis of ethyl mandelate and esterification of alpha-bromopropionic acid by lipase in microemulsions.Chin J Biotechnol.1993,9(1), 33-39.
- Hari M R Gardimalla.; Deendayal Mandal.; Philip D Stevens.; Max Yen.; Yong Gao. Superparamagnetic nanoparticle-supported enzymatic resolution of racemic carboxylates.Chem Commun (Camb).2005,354432-4434.
- Reaction of ɑ-halo alkanoic acids with a dialkyl phosphite in the presence of NaH, followed by treatment of the resulting phosphonoacetate salt with an aldehyde (or cycloalkanone) provides a convenient one-pot alternative to the Knoevenagel or Horner-Wadsworth-Emmons reactions for the synthesis of a variety of substituted acrylic acids: J. Org. Chem., 46, 2514 (1981):
- For a simplified variant of the reaction, applicable to the synthesis of ɑ-unsubstituted cinnamic acids, see Chloroacetic acid, A11482.