Search
Thermo Scientific Chemicals
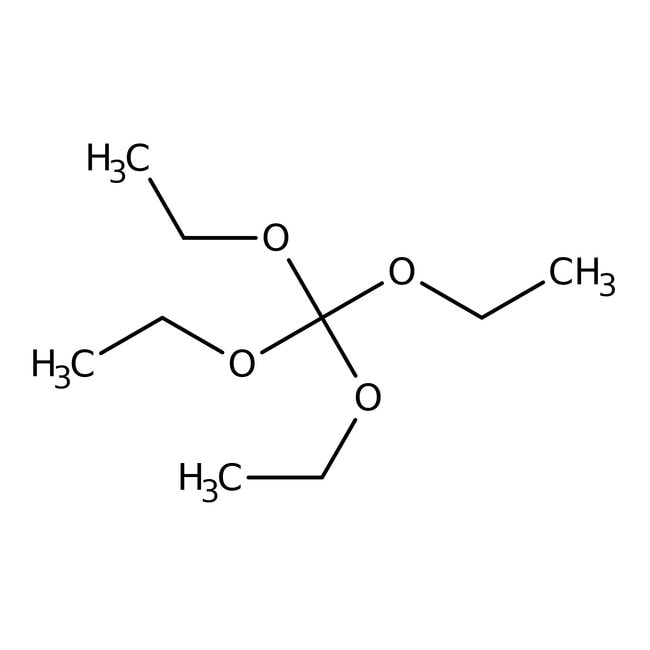
Tetraethyl orthocarbonate, 97+%
CAS: 78-09-1 | C9H20O4 | 192.26 g/mol
Catalog number A17835.22
also known as A17835-22
Price (USD)
463.65
Special offer
Online exclusive
Ends: 30-Jun-2026
545.00Save 81.35 (15%)
Each
Quantity:
100 g
Price (USD)
463.65
Special offer
Online exclusive
Ends: 30-Jun-2026
545.00Save 81.35 (15%)
Each
Chemical Identifiers
CAS78-09-1
Specifications Specification Sheet
Specification Sheet
Appearance (Color)Clear colorless
Assay (GC)≥98.0%
Refractive Index1.3905-1.3945 @ 20?C
Identification (FTIR)Conforms
FormLiquid
Tetraethyl Orthocarbonate is used in the synthesis of chemokine receptor-5 inhibitors against HIV-1, benzobisoxazoles and in the preparation of organic semiconductors. It is also employed in the synthesis of 2,7-dimethylene-1,4,6,9-tetraoxaspiro[4,4]nonane and in the synthesis of cross linked poly(orthocarbonate)s used as organic solvent absorbent.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Tetraethyl Orthocarbonate is used in the synthesis of chemokine receptor-5 inhibitors against HIV-1, benzobisoxazoles and in the preparation of organic semiconductors. It is also employed in the synthesis of 2,7-dimethylene-1,4,6,9-tetraoxaspiro[4,4]nonane and in the synthesis of cross linked poly(orthocarbonate)s used as organic solvent absorbent.
Solubility
Insoluble in water. Contact with water releases a flammable alcohol. Soluble in chloroform and methanol.
Notes
Moisture Sensitive. Store away from oxidizing agents. Keep the container tightly closed and place it in a cool, dry and well ventilated condition. Keep away from ignition sources. Store under inert atmosphere and refrigerated.
Tetraethyl Orthocarbonate is used in the synthesis of chemokine receptor-5 inhibitors against HIV-1, benzobisoxazoles and in the preparation of organic semiconductors. It is also employed in the synthesis of 2,7-dimethylene-1,4,6,9-tetraoxaspiro[4,4]nonane and in the synthesis of cross linked poly(orthocarbonate)s used as organic solvent absorbent.
Solubility
Insoluble in water. Contact with water releases a flammable alcohol. Soluble in chloroform and methanol.
Notes
Moisture Sensitive. Store away from oxidizing agents. Keep the container tightly closed and place it in a cool, dry and well ventilated condition. Keep away from ignition sources. Store under inert atmosphere and refrigerated.
RUO – Research Use Only
General References:
- Tagoshi H and Endo T. Radical polymerization of unsaturated spiroorthocarbonate.J. Polym. Sci. A.,1989,27(4), 1415-1418.
- Sonmez HB, et al. Use of crosslinked poly (orthosilicate) s as organic solvent absorbent.Polymer Prepr.;2008,49(846).
- Wieslaw M. Kazmierski.; Don L. Anderson.; Christopher Aquino.; Brian A. Chauder.; Maosheng Duan.; Robert Ferris.; Terrence Kenakin.; Cecilia S. Koble.; Dan G. Lang.; Maggie S Mcintyre.; Jennifer Peckham.; Christian Watson.; Pat Wheelan.; Andrew Spaltenstein.; Mary B. Wire.; Angilique Svolto.; and Michael Youngman. Novel 4,4-Disubstituted Piperidine-Based C-C Chemokine Receptor-5 Inhibitors with High Potency against Human Immunodeficiency Virus-1 and an Improved human Ether-a-go-go Related Gene (hERG) Profile.J. Med. Chem.,2011,54(11), 3756-3767.
- Used to protect the 2,3-diol system of nucleosides: J. Chem. Soc., Chem. Commun., 552 (1969).
- For a review of the chemistry of orthocarbonates, see: Synthesis, 73 (1977).