Search
Thermo Scientific Chemicals
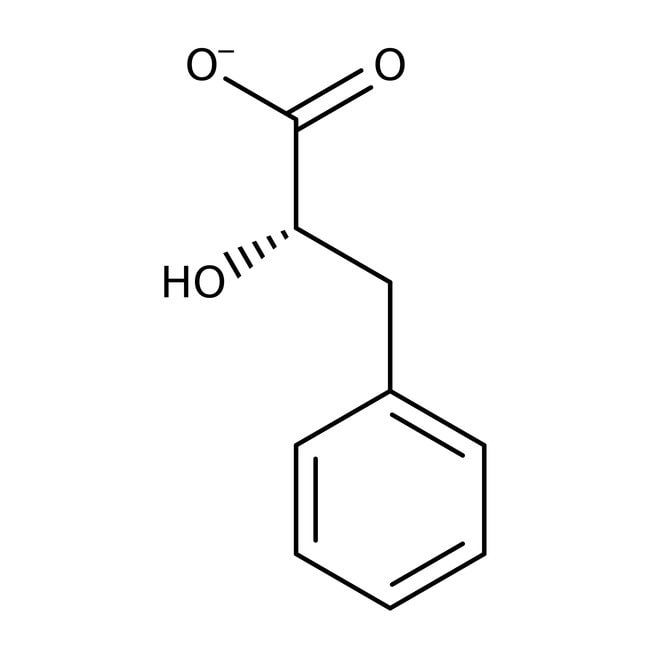
L-(-)-3-Phenyllactic acid, 98%
CAS: 20312-36-1 | C9H9O3 | 165.17 g/mol
Catalog number A18203.14
also known as A18203-14
Price (USD)
346.00
Each
Quantity:
25 g
Price (USD)
346.00
Each
Chemical Identifiers
CAS20312-36-1
IUPAC Name(2S)-2-hydroxy-3-phenylpropanoate
Molecular FormulaC9H9O3
InChI KeyVOXXWSYKYCBWHO-QMMMGPOBSA-M
SMILESO[C@@H](CC1=CC=CC=C1)C([O-])=O
View more
Specifications Specification Sheet
Specification Sheet
Appearance (Color)White to cream or pale yellow
FormCrystals or powder or crystalline powder or needles
Melting Point (clear melt)121-127°C
Optical Rotation-17° to -20° (c = 1 in methanol)
Thin Layer Chromatography≥97.5%
L-(-)-3-Phenyllactic acid is used as pharmaceutical intermediate.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
L-(-)-3-Phenyllactic acid is used as pharmaceutical intermediate.
Solubility
Solubility in water gives very faint turbidity.
Notes
Stable under recommended storage conditions. Incompatible with oxidizing agents.
L-(-)-3-Phenyllactic acid is used as pharmaceutical intermediate.
Solubility
Solubility in water gives very faint turbidity.
Notes
Stable under recommended storage conditions. Incompatible with oxidizing agents.
RUO – Research Use Only
General References:
- G.Gubitz; S.Mihellyes. Direct separation of 2-hydroxy acid enantiomers by high-performance liquid chromatography on chemically bonded chiral phases. Chromatographia. 1984, 19,(1), 257-259.
- Claudia Desiderio; Zeineb Aturki; Dr. Salvatore Fanali. Separation of α-hydroxy acid enantiomers by high performance capillary electrophoresis using copper(II)-L-amino acid and copper(II)-aspartame complexes as chiral selectors in the background electrolyte. Electrophoresis. 1994, 15,(1), 864-869.