Search
Thermo Scientific Chemicals
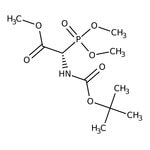
(+/-)-Boc-alpha-phosphonoglycine trimethyl ester, 95%
CAS: 89524-98-1 | C10H20NO7P | 297.24 g/mol
Catalog number B25421.06
also known as B25421-06
Price (USD)
170.65
Special offer
Online exclusive
Ends: 30-Jun-2026
201.00Save 30.35 (15%)
Each
Quantity:
5 g
Price (USD)
170.65
Special offer
Online exclusive
Ends: 30-Jun-2026
201.00Save 30.35 (15%)
Each
Chemical Identifiers
CAS89524-98-1
IUPAC Namemethyl (2S)-2-{[(tert-butoxy)carbonyl]amino}-2-(dimethoxyphosphoryl)acetate
Molecular FormulaC10H20NO7P
InChI KeyLJHAPRKTPAREGO-ZETCQYMHSA-N
SMILESCOC(=O)[C@@H](NC(=O)OC(C)(C)C)P(=O)(OC)OC
View more
Specifications Specification Sheet
Specification Sheet
Appearance (Color)White to cream or pale yellow
Elemental AnalysisC: 40.11 - 40.71% (Theory 40.41%)
Elemental AnalysisH: 6.48 - 7.08% (Theory 6.78%)
Elemental AnalysisN: 4.41 - 5.01% (Theory 4.71%)
Elemental AnalysisP: 9.90 - 10.72% (Theory 10.42%)
View more
It is used as Wittig-Horner reagent for preparing (Z)-Boc-protected dehydroamino acid derivatives and saturated, unnatural amino acids by subsequent asymmetric hydrogenation.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
It is used as Wittig-Horner reagent for preparing (Z)-Boc-protected dehydroamino acid derivatives and saturated, unnatural amino acids by subsequent asymmetric hydrogenation.
Solubility
Insoluble in water.
Notes
Store in cool place. Keep container tightly closed in a dry and well-ventilated place. Incompatible material is strong oxidizing agents.
It is used as Wittig-Horner reagent for preparing (Z)-Boc-protected dehydroamino acid derivatives and saturated, unnatural amino acids by subsequent asymmetric hydrogenation.
Solubility
Insoluble in water.
Notes
Store in cool place. Keep container tightly closed in a dry and well-ventilated place. Incompatible material is strong oxidizing agents.
RUO – Research Use Only
General References:
- Allyn T. Londregan; David Bernhardson; James Bradow; Teresa M. Makowski; Gregory Storer; Joseph Warmus; Ceshea Wooten; Xiaojing Yang. The enantioselective synthesis of (R)- and (S)-3-amino-3,4-dihydro-1H-[1,8]naphthyridin-2-one. Tetrahedron: Asymmetry. 2010, 21 (17), 2072-2075.
- Kåre B. Jørgensen; Odd R. Gautun. Efficient stereoselective preparation of protected isodityrosines. Tetrahedron. 1999, 55 (34), 10527-10536.