Search
Thermo Scientific Chemicals
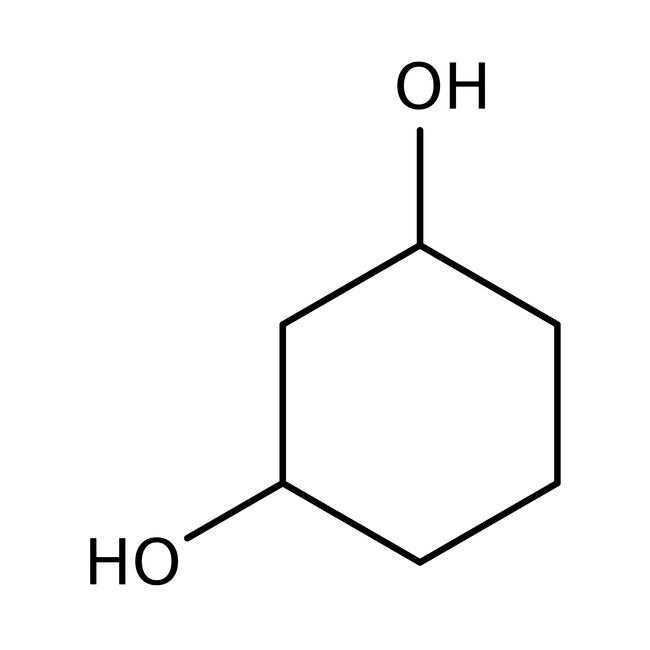
1,3-Cyclohexanediol, cis + trans, 98%
CAS: 504-01-8 | C6H12O2 | 116.16 g/mol
Catalog number L04110.14
also known as L04110-14
Price (USD)
113.65
Special offer
Online exclusive
Ends: 30-Jun-2026
133.00Save 19.35 (15%)
Each
Quantity:
25 g
Price (USD)
113.65
Special offer
Online exclusive
Ends: 30-Jun-2026
133.00Save 19.35 (15%)
Each
Chemical Identifiers
CAS504-01-8
IUPAC Namecyclohexane-1,3-diol
Molecular FormulaC6H12O2
InChI KeyRLMGYIOTPQVQJR-UHFFFAOYNA-N
SMILESOC1CCCC(O)C1
View more
Specifications Specification Sheet
Specification Sheet
Assay (Silylated GC)≥97.5%
Appearance (Color)Colorless to white
FormCrystals or powder or crystalline powder or fused solid or waxy solid or clear liquid or viscous liquid as melt
Identification (FTIR)Conforms
1,3-Cyclohexanediol is an important raw material and intermediate used in organic synthesis, pharmaceuticals, agrochemicals and dyestuff fields.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
1,3-Cyclohexanediol is an important raw material and intermediate used in organic synthesis, pharmaceuticals, agrochemicals and dyestuff fields.
Solubility
Soluble in water.
Notes
Keep container tightly sealed. Store in cool, dry conditions in well sealed containers. Incompatible with oxidizing agents.
1,3-Cyclohexanediol is an important raw material and intermediate used in organic synthesis, pharmaceuticals, agrochemicals and dyestuff fields.
Solubility
Soluble in water.
Notes
Keep container tightly sealed. Store in cool, dry conditions in well sealed containers. Incompatible with oxidizing agents.
RUO – Research Use Only
General References:
- J. Winkler.; F.W. McLafferty. Stereochemical effects in the chemical ionization mass spectra of cyclic diols. Tetrahedron. 1974, 30 (17), 2971-2976.
- John T. Groves.; Michael Van der Puy. Stereospecific aliphatic hydroxylation by an iron-based oxidant. J. Am. Chem. Soc. 1974, 96 (16), 5274-5275.