Search
Thermo Scientific Chemicals
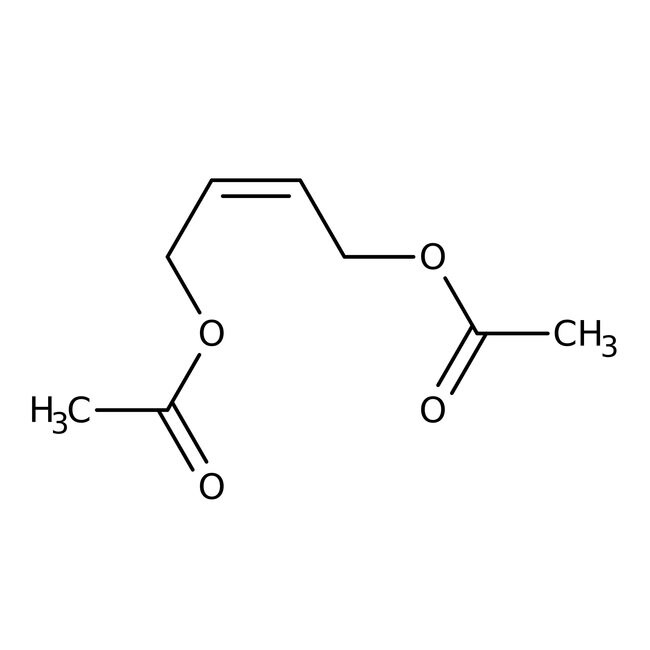
cis-1,4-Diacetoxy-2-butene, 96%
CAS: 25260-60-0 | C8H12O4 | 172.18 g/mol
Catalog number L13024.09
also known as L13024-09
Price (USD)
50.50
Each
Quantity:
10 g
Price (USD)
50.50
Each
Chemical Identifiers
CAS25260-60-0
IUPAC Name(2Z)-4-(acetyloxy)but-2-en-1-yl acetate
Molecular FormulaC8H12O4
InChI KeyVZUAUHWZIKOMFC-ARJAWSKDSA-N
SMILESCC(=O)OC\C=C/COC(C)=O
View more
Specifications Specification Sheet
Specification Sheet
Appearance (Color)Clear colorless to yellow
Assay (GC)≥94.0%
Refractive Index1.4390-1.4465 @ 20°C
FormLiquid
cis-1,4-Diacetoxy-2-butene is used as an organic chemical synthesis intermediate.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
cis-1,4-Diacetoxy-2-butene is used as an organic chemical synthesis intermediate.
Solubility
Not miscible in water.
Notes
Stable under recommended storage conditions. Incompatible with oxidizing agents.
cis-1,4-Diacetoxy-2-butene is used as an organic chemical synthesis intermediate.
Solubility
Not miscible in water.
Notes
Stable under recommended storage conditions. Incompatible with oxidizing agents.
RUO – Research Use Only
General References:
- Shyh-Chyun Yang; Pei-Chin Liu; Wei-Hao Feng. Palladium-catalyzed tandem allylation of 1,2-phenylenediamines with cis-1,4-diacetoxy-2-butene. Tetrahedron Letters. 2004, 45,(25), 4951-4954.
- Jacob M. Berlin; Steven D.Goldberg Dr.; Robert H.Grubbs Prof. Highly active chiral ruthenium catalysts for asymmetric cross- and ring-opening cross-metathesis. Angewandte Chemie. 2006, 118,(45), 7753-7757.