Search Thermo Fisher Scientific
Invitrogen
Goat anti-Rat IgG (H+L) Cross-Adsorbed Secondary Antibody, Alexa Fluor™ 350
FIGURE: 1 / 1
Rat IgG (H+L) Cross-Adsorbed Secondary Antibody (A-21093) in IHC (F)

Product Details
A-21093
Immunohistochemistry (Frozen) (IHC (F))
Immunocytochemistry (ICC/IF)
Species Reactivity
Host/Isotype
Class
Type
Immunogen
Conjugate
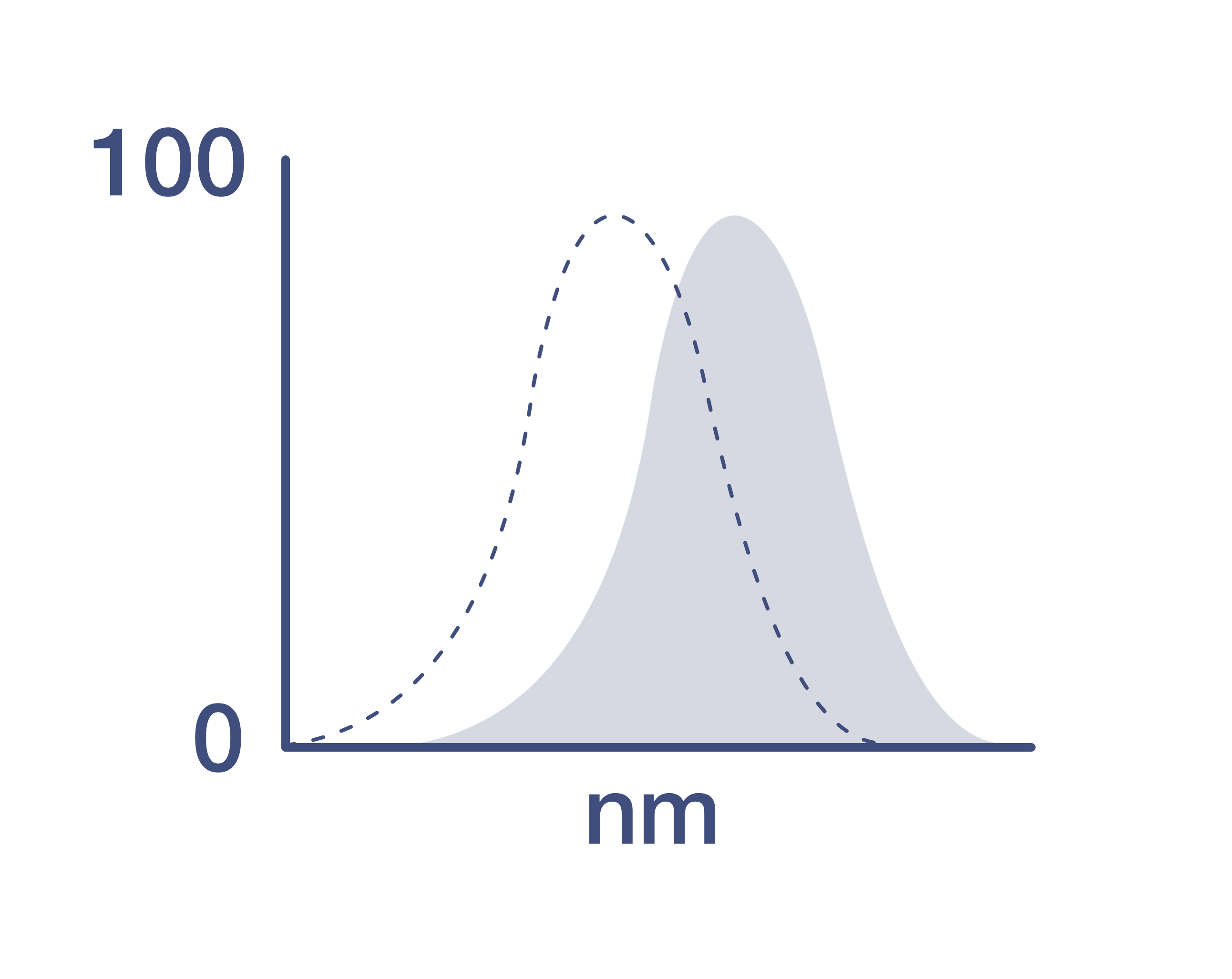
Excitation/Emission Max
Form
Concentration
Purification
Storage buffer
Contains
Storage conditions
Shipping conditions
RRID
Target
Cross Adsorption
Antibody Form
Product Specific Information
Product will be shipped at Room Temperature.
Target Information
Anti-Rat secondary antibodies are affinity-purified antibodies with well-characterized specificity for rat immunoglobulins and are useful in the detection, sorting or purification of its specified target. Secondary antibodies offer increased versatility enabling users to use many detection systems (e.g. HRP, AP, fluorescence). They can also provide greater sensitivity through signal amplification as multiple secondary antibodies can bind to a single primary antibody. Most commonly, secondary antibodies are generated by immunizing the host animal with a pooled population of immunoglobulins from the target species and can be further purified and modified (i.e. immunoaffinity chromatography, antibody fragmentation, label conjugation, etc.) to generate highly specific reagents.
For Research Use Only. Not for use in diagnostic procedures. Not for resale without express authorization.

Performance Guarantee
If an Invitrogen™ antibody doesn't perform as described on our website or datasheet,we'll replace the product at no cost to you, or provide you with a credit for a future purchase.*
Learn more
We're here to help
Get expert recommendations for common problems or connect directly with an on staff expert for technical assistance related to applications, equipment and general product use.
Contact tech support