Search
NMR Tech Talk: April 2014
In this issue
Many people want to learn whether a bench top NMR instrument is suitable for them. An application chemist's evaluation process includes asking about their workflow, their present NMR access and usage, and the typical sample types that they want to analyse by NMR. The prediction tools in Mnova™ are extremely helpful to show show the sort of results people can expect from their samples at an early stage, before planning a site visit. This works well for 'small molecule' people; typically synthetic organic chemists working on drug like molecules with a molecular weight of around 500 Da.
The purpose of this experiment is to perform a microscale synthesis of acetylferrocene from ferrocene and acetic anhydride in an acid-catalyzed (85% H3PO4) Friedel-Crafts reaction. The crude reaction product is isolated by filtration and purified on a silica gel column using flash chromatography. Two fractions will be collected from the column separation of the mixture. Ferrocene (fraction 1) is eluted first using hexanes; it appears as a yellow band in the column and represents excess reagent. The product, acetylferrocene (fraction 2), is eluted next using a 50:50 mixture of hexanes and diethyl ether solution. Solutions of pure ferrocene and acetylferrocene, crude isolated product and column purified reaction product will be prepared and analyzed using the Thermo Scientific picoSpin 45 NMR spectrometer.
For those of us who are scientists, we know science stole our hearts, we undoubtedly love science, and we have a curiosity to understand why and discover new things. We are fortunate to do what we love. If we think back, there was a tipping point along our paths that fostered our interest and love of science. Perhaps we attribute our science career to mentors who provided us the opportunity and freedom to explore our curiosity as an undergraduate student? Well, science is stealing another heart: meet fellow scientist in-training, Lilian (Lily) White.
The purpose of this experiment is to separate the components of a mixture using column chromatography. The sample is a 50:50 mixture of ferrocene and acetylferrocene. Two fractions will be collected from the column separation of the mixture. Ferrocene (fraction 1) is eluted first using hexanes and appears as a yellow band in the column. Acetylferrocene (fraction 2) is eluted next using a 50:50 mixture of hexanes and diethyl ether solution.
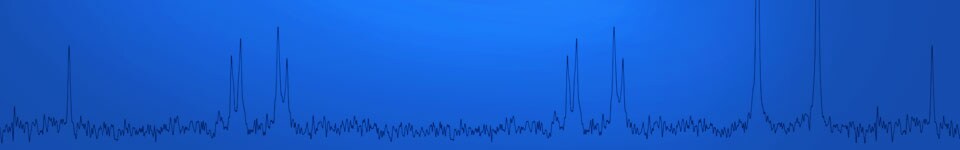
In this experiment, we monitor changes occurring during the course of a simple reaction, the hydrolysis of acetic anhydride with heavy water (D2O) by a modified in situ reaction monitoring technique. We also take advantage of isotopic substitution to suppress an otherwise large proton signal in the NMR spectrum originating from the reactant/solvent H2O. Isotopic substitution does not alter the potential energy surface along the reaction coordinate, but it will affect the rate of reaction by changing the enthalpy of activation.
About NMR Tech Talk
Featuring the latest news, events, and educational approaches in benchtop NMR, Tech Talk is your forum for bringing this interesting and valuable technique into the classroom or as part of your analytical laboratory. Discover what's new from peers and from our experts at Thermo Fisher Scientific.
Spectroscopy, Elemental & Isotope Analysis Resource Library
Access a targeted collection of application notes, case studies, videos, webinars and white papers covering a range of applications for Fourier transform infrared (FTIR) spectroscopy, near infrared spectroscopy, Raman spectroscopy, nuclear magnetic resonance, ultraviolet-visible (UV-Vis) spectrophotometry, X-ray fluorescence, and more.
