Search
Interferon (IFN) Cell Signaling Pathway
The interferon (IFN) pathway plays a critical role in the human immune response. Following viral infection, the human body triggers a complex regulatory system of innate and adaptive immune responses designed to defend against the virus. One of the many responses to the viral invasion is the induction of the pleiotropic cytokines, interferon (IFN) [1]. Induction of IFN gene expression leads to increased cellular resistance to viral infection and may also affect cell growth. IFN is used in the treatment of multiple sclerosis, many types of cancer, hepatitis B and C, and herpes simplex – to name a few [9, 10, 11, 12]. We offer a wide range of products to help with IFN research.
Key interferon (IFN) cell signaling pathway targets
Two subtypes of IFN comprise the IFN family of cytokines:
Type I IFN (IFNAR)
- Over 20 variants identified, including IFNα and IFNβ. All share the ability to bind to IFNAR receptors.
- Have subunits IFNR1 and IFNR2
Type II IFN (IFNGR)
- Only 1 variant – IFN-γ – and binds to the IFN-γ receptor.
- Have subunits IFN-γR1 and IFN-γR2
Activation of the toll-like receptor pathway following viral infection leads to the increased production of IFNα and IFNβ and the further induction of adaptive immune responses by increasing MHC-I (major histocompatibility complex class-I).
To mediate signaling, subunits of the IFN receptors are associated with a member of the JAK-STAT (janus activated kinase-signal transducer and activator of transcription) family:
The IFNR1 subunit is associated with TYK2 (Tyrosine Kinase-2), and IFNR2 is associated with JAK1 [3, 4, 5]. The IFN-γR1 subunit is associated with JAK1 and IFN-γR2 is associated with JAK2 [4]. Activation of both Type I and type II receptors, following ligand binding, results in dimerization and rearrangement of receptor subunits leading to the activation of associated JAKs by auto phosphorylation and the further activation of the STAT proteins. Following phosphorylation, activated STAT forms homodimers, then translocate to the nucleus where they initiate transcription of IFN-stimulated genes [4].
Additional targets IFN acts upon:
Type I IFN can also induce the formation of the ISGF3 complex, composed of STAT1, STAT2, and IRF9. Another important transcriptional complex that is induced by Type-I IFN is the ISGF3 (ISG factor-3) Complex [6, 7]. The ISGF3 complexes bind ISRE (IFN-stimulated response elements) further inducing the transcription of IFN-stimulated genes which contain ISREs within their promoters.
IFNγ binding to the IFNγ-receptor leads to the downstream tyrosine phosphorylation of STAT1 at residue Tyr701. The resulting Tyr701-phosphoryled STAT1 homodimers translocated to the nucleus and bind to GAS (IFNγ-activated sites) elements to induce expression of IFNγ-regulated genes. [6].
The mitogen activated protein kinase (MAPK) pathway has also been shown to be regulated by IFN signaling [8]. Activated JAKs phosphorylate Vav, a guanine nucleotide exchange factor, resulting in the downstream activation of Rac1. Activated Rac1 further activates MEKK1 (MAP3K1), leading to the phosphorylation of MEK3 (MAP2K3) and MEK6 (MAP2K6) which in turn regulates p38MAPK (MAPK14) phosphorylation. Activated p38 subsequently regulates activation of multiple downstream effectors, including the mitogen-and stress-activated kinases MSK1 and MSK2. The specific transcription factors that are regulated by p38s include CREB (cAMP responsive element binding protein) and histone-H3.
Activated TYK2 and JAK1 regulate tyrosine phosphorylation of IRS1 and IRS2 (insulin receptor substrate), which provide docking sites for the SH2 (SRC homology-2) domains of the regulatory subunit (p85) of PI3K (phosphatidylinositol 3-kinase). PI3K further activates mTOR (mammalian target of rapamycin), a critical regulator of translational proteins.
Data
We offer antibodies, ELISAs, Luminex multiplex assays and growth factors for key targets in the interferon (IFN) cell signaling pathway. For more information please visit our Life Sciences Home Page.
Featured below is flow cytometry and ELISA data using Thermo Scientific products.
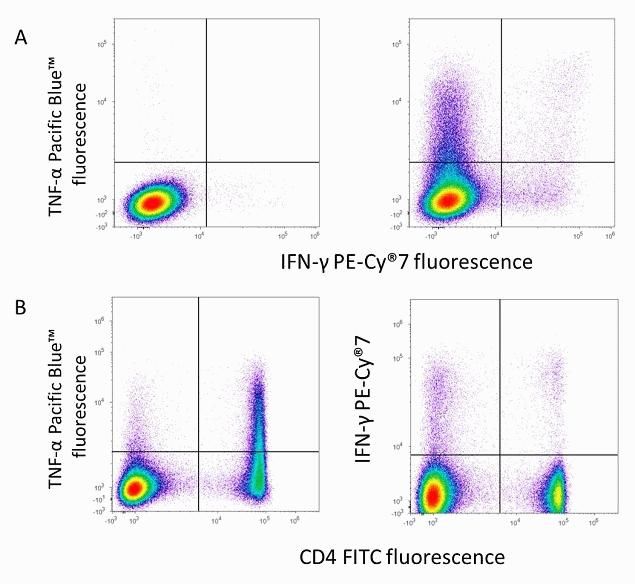
Flow cytometry analysis of IFN-gamma on C57BL/6 mouse splenocytes. Cell were left unstimulated (left panels) or stimulated (right panels) for 5 hours with phorbol myristate acetate (PMA) and ionomycin in the presence of brefeldin A. Cells were surface-stained with anti-mouse CD4 FITC (Cat. No. MCD0401). Then, cells were fixed and permeabilized using FIX & PERM reagents. Cells were stained intracellularly with anti-mouse interferon gamma (IFN-gamma) PE-Cy7 conjugate (Cat. No. A18713) and anti-mouse tumor necrosis factor alpha (TNF-a) Pacific Blue conjugate (Cat. No. RM90128).
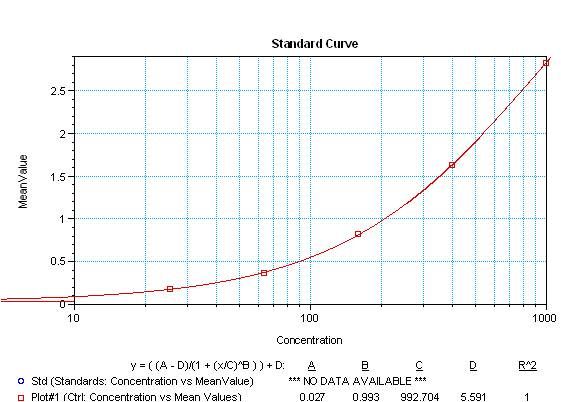
Sandwich ELISA analysis of IFN-gamma was performed using the Thermo Scientific ELISA kit EHINFG by loading 50 µL per well of interferon gamma recombinant protein (Cat. No. SIFNG) in quadruplicate at 1000, 400, 160, 64, 25.6 and 0 pg/mL across a 3 µg/mL mouse anti-human IFNG monoclonal antibody (Cat. No. M700A) pre-coated plate and incubated for 2 hours at room temperature. The plate was washed then incubated with 50 µL per well of a mouse anti-human IFNG biotinylated antibody in all test wells at 1:40,000 for 30 minutes at room temperature. Detection was performed using 1-Step Ultra TMB Substrate (Cat. No. 34028) for 30 minutes at room temperature in the dark. The plate was then stopped with 0.16M sulfuric acid. Absorbances were read on a spectrophotometer at 450-550 nm.
References
- Barber G.N. (2001) Host defense, viruses and apoptosis. Cell Death Differ. 8(2) 113-26.
- Mahajan B.B. et al (2015) Interferons. Indian J Dermatol Venereol Leprol 81: 51-55.
- Nguyen K.B. et al. (2000) Interferon α/β-mediated inhibition and promotion of interferon γ: STAT1 resolves a paradox. Nat Immunol. 1: 70-6.
- Chen, J. et al. (2004) Diversity and relatedness among the type I interferons. J Interferon Cytokine Res. 24: 687-698.
- Katsoulidis, E. et al. (2005) The p38 mitogen-activated protein kinase pathway in interferon signal transduction. J Interferon Cytokine Res. 25:749-56.
- Aaronson, D.S. et al. (2002) A road map for those who don’t know JAK-STAT. Science 296:1653-1655.
- Platanias, L.C. (2005) Mechanisms of type-I and type-II interferon mediated signalling. Nat. Rev. Immunol. 5:375-376.
- David, M. (2002) Signal transduction by type I interferons. BioTechniques Suppl: 58-65.
- Paolicelli, D. et al. (2009) Review of interferon beta-1b in the treatment of early and relapsing multiple sclerosis. Biologics 3: 369-376.
- Goldstein, D. et al. (2008) The role of interferon in cancer therapy: a current perspective. CA: A Cancer Journal for Clinicians 5: 258-277.
- Cooksley, W.G. (2004) The role of interferon therapy in hepatitis B. Gastroenterology Expert Column. 6: 6.
- Wilhelmus, K.R. (2010) Antiviral treatment and other therapeutic interventions for herpes simplex virus epithelial keratitis. Cochrane Database Syst Rev. 12.
- Ivashkiv, L.B. et al. (2014) Regulation of type I interferon McNab, F. et al (2015) Type I interferons in infectious disease. Nature Reviews Immunology 15: 87-103.
- McNab, F. et al (2015) Type I interferons in infectious disease. Nature Reviews Immunology 15: 87-103.
- Gonzalez-Navajas, J.M. et al (2012) Immunomodulatory functions of type I interferons. Nature Reviews Immunology 12: 125-135.
Featured products
| Product name | Cat. No. |
| IFN gamma Antibody | M700A |
| STAT5b Antibody | 135300 |
| IFN alpha Antibody | M710 |
| STAT3 Antibody | MA113042 |
| Phospho-STAT1 pTyr701 Antibody | 333400 |
| Phospho-PI3K p85 pTyr458+p55 pTyr199 Antibody | PA517387 |
| JAK2 Antibody | AHO1352 |
| 44422G | |
| PA517697 | |
| IRF9 Antibody | PA530378 |
| PA14703 | |
| PA517898 | |
| mTOR Antibody | PA534663 |
| 214051 | |
| IRS2 Antibody | PA514897 |
| PA515085 | |
| p38 Antibody | PA527831 |
| STAT2 Antibody | PA534683 |
| PA14619 | |
| GAS6 Antibody | PA528227 |