Search
Pipette Accuracy vs. Precision

Accurate Data. More Discoveries.
- Forward, Reverse, Repetitive, & Heterogeneous Sample Pipetting
- Pipetting Recommendations for Different Sample Types
- 10 Steps to Improve Pipetting Accuracy
- Preventing Cross Contamination
Complete the form and download a pdf version of the GLP Guide.
Accuracy
Accuracy is the quality of being true, correct, exact, and free from error. Accuracy is the ability of a pipette to give a response close to a true or nominal volume as indicated by the volume setting.
Precision
Precision is often referred to as repeatability or sample reproducibility, and also as a standard deviation.
Error-free pipetting requires both precision and accuracy.
When pipettes are both accurate and precise the mean volume is the set volume and there is no variation between different pipettings.
Example: The pipette volume is set at 20 µl
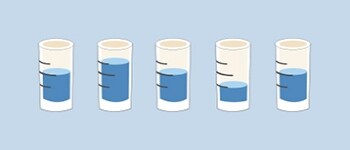
Accurate, but not precise:
The mean volume is the correct (set) volume, but the separate pipettings differ from the set volume.
Precise, but not accurate:
There is no variation between the separate pipettings, but the mean volume differs from the set volume.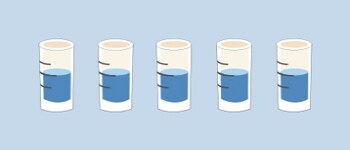
Accurate and precise:
The mean volume is the set volume, and there is no variation between the different pipettings.Factors Affecting Pipette Accuracy

Temperature
Temperature has many effects on pipetting accuracy. The factor that has the greatest effect is the temperature difference between the delivery device and the liquid. The air gap (dead air volume) between the liquid surface and the piston experiences thermal expansion effects unique to the case. This either reduces or increases the liquid amount aspirated into the tip, along with other effects.

Density
The density (mass/volume ratio) affects the liquid volume that is aspirated into the tip. A smaller dose of liquid with higher density than water is aspirated compared to a similar operation with water. With lower density liquids the effect is the opposite. This is caused by the flexible dead air volume along with earth’s gravity. The density of liquids also varies according to temperature. Typically the density of liquids also varies according to temperature. Typically the density for water is 0.998 kg/dm3, for ethanol 0.79 kg/dm3, and for sulfuric acid (95–98% h2SO4) 1.84 kg/dm3 (the values apply at the temperature of 20 °C/68 °F).

Altitude
Geographic altitude affects accuracy through air pressure. Air pressure decreases in higher altitudes, and the conversion factor Z decreases as well. Also, with some liquids, the boiling point decreases quite close to room temperature, which will increase the evaporation loss dramatically.
California Proposition 65 Warning: Products manufactured with polycarbonate (PC), polyethylene terephthalate (PET), polyethylene terephthalate glycol (PETG) or polystyrene (PS) contain chemicals known to the State of California to cause cancer, birth defects or other reproductive harm.



