Search
Detection of Tumor Markers—IHC Solutions

Immunohistochemistry using Invitrogen antibodies: Superior performance through advanced verification
From sample prep through image acquisition, Thermo Fisher Scientific offers a broad portfolio of superior reagents, accessories, and instruments to help with every step of the immunohistochemistry (IHC) workflow (Figure 1).

Figure 1. Overview of the IHC process.
One of the critical reagents for a successful IHC experiment is the detection antibody. We are striving to provide superior antibody performance by testing the specificity of Invitrogen antibodies in accordance with the newly adopted advanced verification testing methods for antibody validation.* To help ensure superior antibody results, we’ve expanded our specificity testing methodology using a two-part approach for advanced verification.
Part 1—Target specificity verification: verifies the antibody will bind to the correct target. Our antibodies are being tested using at least one of our specificity tests to ensure proper functionality in researchers’ experiments.
Part 2—Functional application validation: verifies the antibody works in a particular application(s) of interest.
Tumor detection
Cancer protein biomarkers are useful in applications such as IHC to identify where and to what degree cancer has affected healthy tissues. These proteins can be expressed in some cell or tissue types but not in healthy tissues. Antibody target verification can be determined by analyzing the relative expression of these proteins in different tissue types using IHC. For example, the tumor suppressor p53 protein is known to be strongly expressed in the nuclei of tumor cells within ovarian cancer tissue, compared to low expression seen in normal, healthy ovary tissue. Additionally, p53 expression is not detectable in other tissues, such as normal, healthy human liver. The IHC analysis of p53 expression in positive ovarian cancer and negative ovary and liver tissues shown in Figure 2 demonstrates the specificity of the p53 antibody used for the detection. As expected, p53 is highly expressed (stained brown) in the nuclei of ovarian cancer cells, but expressed at a low level in normal ovary tissue and not expressed in normal liver tissue.
The same antibody verification and IHC workflow described can also be performed using immunofluorescence (IF). For fluorescence detection, the primary or secondary antibody is conjugated to a fluorophore that is detected by fluorescent microscopy. Recently introduced, Invitrogen Alexa Fluor Plus secondary antibodies offer high signal-to-noise ratios and superior brightness. Figure 3 depicts an example of IHC detection by IF using a p53 monoclonal primary antibody and an Alexa Fluor Plus 488 conjugated secondary antibody to visualize the relative expression of p53.
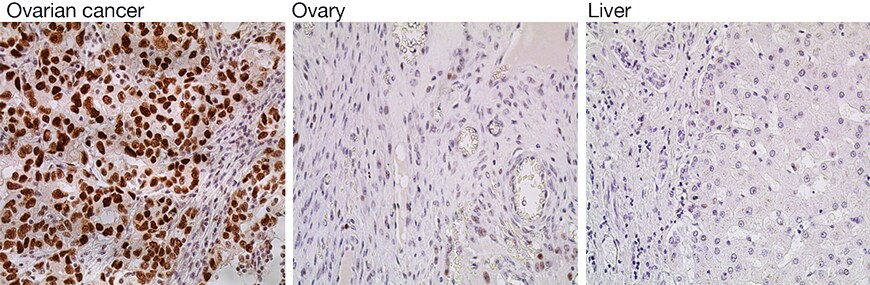
Figure 2. Detection of relative p53 expression by IHC. The specificity of anti-p53 monoclonal antibody (Cat. No. MA5-12571) was demonstrated by detecting relative p53 expression in ovarian cancer tissue (positive, left panel), normal ovary tissue (low expression, middle panel), and normal human liver tissue (negative, right panel). Detection was performed using a goat anti-mouse IgG (H+L) secondary antibody and Alexa Fluor Plus 488 conjugate (Cat. No. A32723). Tissues were imaged on the Invitrogen EVOS FL Auto Imaging Station.

Figure 3. IHC detection of p53 across tissues by immunofluorescence. The specificity of anti-p53 monoclonal antibody (Cat. No. AHO0152) was demonstrated by detecting relative p53 expression in ovarian cancer tissue (positive, left panel), normal ovary tissue (low expression, middle panel), and normal human liver tissue (negative, right panel). Images were taken on an EVOS FL Auto Imaging Station.
For Research Use Only. Not for use in diagnostic procedures.