Search
Physical Properties that Define Fluorescence

Fluorescence is a function of light energy
Fluorescent molecules by definition absorb light at one color (wavelength) and emit it at another. The difference in colors is called the Stokes shift. The cameras used in fluorescence microscopy allow the detection of signal beyond the wavelengths our eyes can see.
Learn about the physical properties that define fluorescence, including wavelength, how energy relates to fluorescence and fluorescent colors, and what defines a fluorescent molecule's spectra.
The visible spectrum of light
When we talk about light in microscopy, it is usually noted as wavelength, even though photons (the packets of energy that make light) can act as both particles and waves. Visible light, or light that we can see with our eyes, is usually in the range of 400–700 nm and encompasses all colors in the rainbow, with blues starting at around 400 nm and reds finishing at around 700 nm.
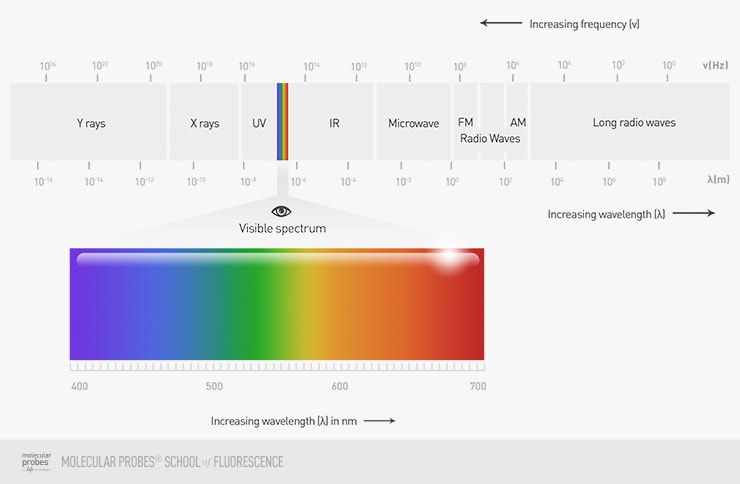
Figure 1. The electromagnetic spectrum, with visible wavelengths and their corresponding colors highlighted.
Range of detection in fluorescence microscopy
 | The range for fluorescence imaging extends a bit beyond where our eyes can see. Using this extended range is no problem because the CCD cameras that collect light emitted from our sample in a typical fluorescence microscopy setup have a wider range than our eyes do. In practical terms, imaging wavelengths for cell biology are usually in the range of 300–800 nm. Figure 2. The range and efficiency of light detection for a CCD camera compared to that of the human eye. |
What is fluorescence?
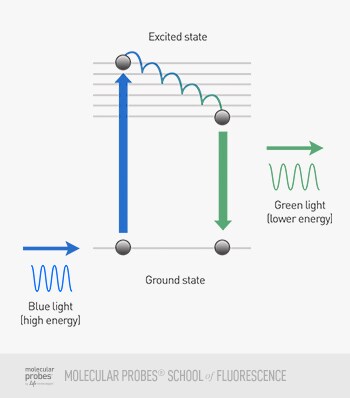
But what is special about the term fluorescence? Fluorescence refers to the physical property of an object absorbing light at one wavelength and then reemitting it at another wavelength. If a molecule absorbs the light of one wavelength and emits it in another (i.e., fluoresces), we call that molecule a fluorophore. Usually the wavelength the molecule emits will be lower energy than what it absorbed, so in simpler terms we could say that something absorbs blue light and emits green, or absorbs green light and emits red. Further study:Watch "Introduction to Fluorescence" Figure 3. The inverse relationship between energy and wavelength in the visible spectrum. |  |
Figure 4. Simplified Jablonski diagram showing the energy state change of a fluorophore’s electron as it undergoes fluorescence, with the corresponding change in the color of light | Where the fluorescent signal comes fromTo understand this at a deeper level, we need to think about photons, the packets of energy that make up light. The magnitude of energy that a photon contains determines its color or, in physical terms, its wavelength. When the light (or photon) hits a fluorophore, the energy is transferred to the fluorophore’s electrons. The electrons are excited, but then rapidly lose that extra energy (that sounds like people too, doesn’t it?). The end result of this loss of energy is the emission of a photon of light, but that photon will have less energy than the original photon, so it will have a longer wavelength and be a different color. The emitted photons are the signal you need to collect as data during your imaging experiment. |
Excitation and emission spectra
Most fluorophores don’t just absorb light at one discrete wavelength and emit light at another discrete wavelength: they usually absorb and emit a range of wavelengths. So when we think about using fluorophores in imaging, it’s useful to also think about the entire spectrum of their absorption and emission, while at the same time keeping in mind the maximum excitation and emission wavelengths. The maximum values are the peaks of the excitation and emission spectra.
Further study: Watch "Anatomy of Fluorescence Spectra"
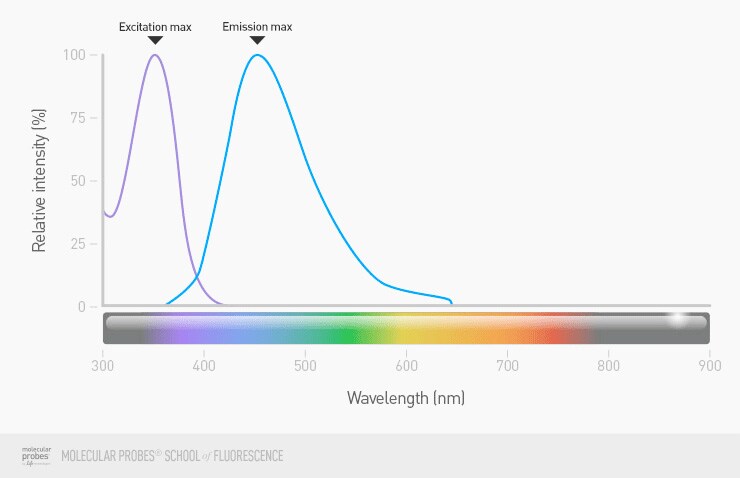
Figure 5. Excitation and emission spectra of a nuclear dye (DAPI). Shows both the fraction of light absorbed by the dye over a range of wavelengths (excitation, shown in purple) as well as the light emitted from the dye over a range of wavelengths (emission, shown in blue).
The all-important Stokes shift
The difference between the excitation and emission maxima for a given fluorophore is called the Stokes shift. A fluorophore with a large Stokes shift will be much easier to use in your imaging then a fluorophore with a small Stokes shift. When there is only a small difference in wavelength between excitation and emission, it will be very difficult for you to see the emitted light from your labeled object as separate from the light used for excitation, and there will be more problems with background fluorescence.
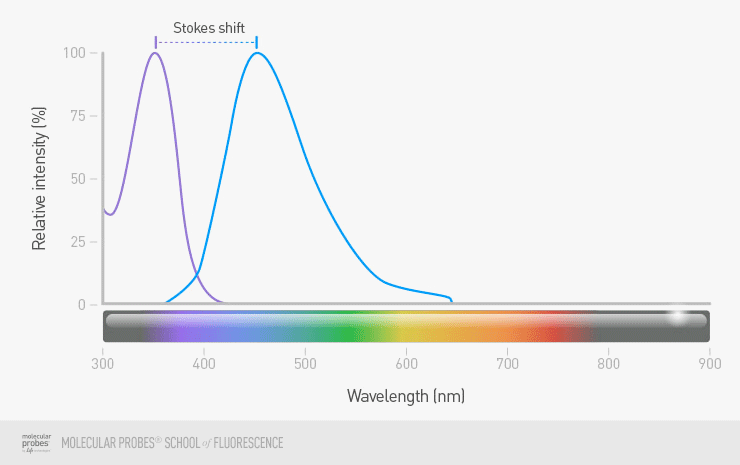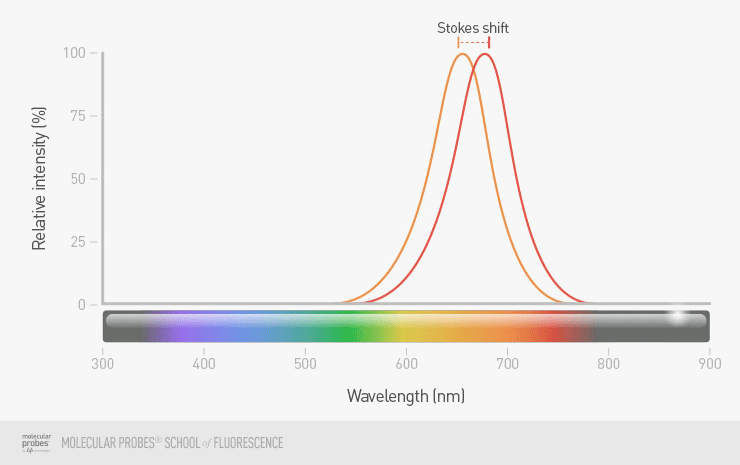
Figure 6. A fluorophore with good separation between the excitation and emission maxima typically results in more reliable detection than a fluorophore with little separation. Compare the fluorophore with a large Stokes shift (purple and blue maximum peaks) to that of a fluorophore with a small Stokes shift (orange and red peaks).
Introduction to Fluorescence
This video provides an easy to understand overview of the basic principles of fluorescence and is suitable for beginners or for those that need a quick refresh.
Anatomy of Fluorescence Spectra
This video describes the principle behind fluorescence spectra and how they can be used to determine properties of a fluorescent molecule.
For Research Use Only. Not for use in diagnostic procedures.