Search
Single Stranded Templates for Pyrosequencing™
The PSQ 96 system from Pyrosequencing™ is used to analyze large numbers of short to medium length DNA sequences.
This widely applicable sequencing technique (1) was developed at the Royal Institute of Technology (Stockholm, Sweden) and commercialized in 1999 by Pyrosequencing AB (now Biotage AB). The strength of their patented technology lies in the rapid generation of sequence data with high accuracy (>99%) and reproducibility (>99%). Real time sequencing by synthesis means there is no need for labeled primers, labeled nucleotides or gel electrophoresis. The whole process from sample preparation to ready-analyzed results is completed in under 2 hours and is easily automated.
Principle of real time sequencing
In a single tube, the nucleotide composition of a growing DNA strand is determined by an enzyme cascade system (Figure 1). Four different enzymes are used—DNA polymerase, ATP sulfurylase, firefly luciferase and apyrase.
The single-stranded DNA template is first hybridized with the sequencing primer and mixed with the enzymes along with the two substrates adenosine 5’phosphosulfate (APS) and luciferin. One of the four nucleotides (dNTP) is then added to the reaction. If the nucleotide is complementary to the base in the template strand, DNA polymerase incorporates it into the growing strand. Pyrophosphate (PPi)—in an amount equal in molarity to that of the incorporated nucleotide—is released and converted to ATP by sulfurylase in the presence of APS.
Detection is based on the amount of visible light produced during the following ATP-mediated conversion of luciferin to oxyluciferin by the luciferase. Any unincorporated nucleotides are degraded by apyrase, before the next nucleotide is added to the tube and the cascade of reactions repeated. Light signals are detected on a Pyrogram™ (Figure 2), on which each peak corresponds to one or more identical bases added to the growing DNA strand.
Preparing the single stranded DNA template
Pyrosequencing™ requires a single stranded sequencing template. The simplest way to prepare this is by the use of Dynabeads M-280 Streptavidin (Figure 3). First the DNA fragment of interest is PCR amplified with one of the primers biotinylated. The amplicons are then immobilized onto the beads and the non-biotinylated strand denatured with NaOH. After strand elution and subsequent washings to remove all other reaction components, the sequencing primer is added and annealed to the pure single stranded DNA template. (Large volume discounts are available, please inquire.)
Simultaneous manual sample preparation can be done for up to 96 samples by using a magnet specially designed to hold 96-well plates. Alternatively, protocols for automated sample preparation with Dynabeads M-280 Streptavidin have been developed with first-class results on the Tecan Genesis Workstation 150 using the Tecan MagS station (TECAN AG, Switzerland) (Figure 2 and Figure 4), and on the Magnatrix 1200 workstation (Magnetic Biosolutions AB, Sweden).
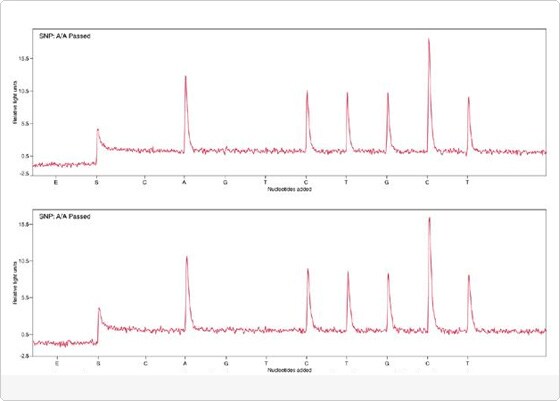
Fig 4. Pyrograms produced with automated (above) and manual (below) template preparation for Pyrosequencing™ SNPs Analysis
Applications of real time sequencing
Pyrosequencing™ was first applied to SNPs analysis in a variety of research areas (3). Since its inception, numerous other applications have been developed.
Short to medium DNA sequences (typically 20-30 bases) are quickly obtained, making real time sequencing technology from Pyrosequencing™ a useful technique for the identification of disease-related mutations (4) and analysis of genomic diversity (5). Genotyping of infectious agents is another area where the technology has been applied for sequence analysis.
Successful typing of human papillomavirus (6) and Helicobacter pylori (7) has recently been reported. Within the area of disease management, Pyrosequencing™ technology has been used to monitor drug therapy by detecting resistance to HIV-1 protease inhibitors (8).
References
- Ronaghi M, Uhlén M and Nyrén P. A Sequencing Method Based on Real-Time Pyrophosphate. Science 1998;281:363-365.
- Blomgren P et al. Manual and automatic sample preparation for Pyrosequencing. Pyrosequencing AB poster.
- Alderborn A, Kristofferson A and Hammerling U. Determination of single-nucleotide polymorphisms by real-time pyrophosphate DNA sequencing. Genome Res. 2000;10(8):1249-1258.
- Ahmadian A et al. Analysis of the p53 tumor suppressor gene by pyrosequencing. Biotechniques 2000;28(1):140-144.
- Schiller AL et al. Accurate Allele Frequency Estimations of SNPs using Pyrosequencing. Pyrosequencing AB poster.
- Gharizadeh B et al. Typing of human papillomavirus by pyrosequencing. Lab. Invest. 2001;81(5):673-679.
- Monstein H, Nikpour-Badr S and Jonasson J. Rapid molecular identification and subtyping of Helicobacter pylori by pyrosequencing of the 16S rDNA variable V1and V3 regions. FEMS Microbiol. Lett. 2001;199(1):103-107.
- O’Meara D et al. Monitoring resistance to human immunodeficiency virus type 1 protease inhibitors by pyrosequencing. J. Clin. Microbiol. 2001;39(2):464-473.
Figures 1, 2 and 4 are a courtesy of Pyrosequencing AB. Pyrosequencing™ and Pyrogram™ are trademarks of Pyrosequencing AB, Uppsala, Sweden.


