Search
miRNA Biogenesis
RNA polymerase II transcribes miRNA genes, generating long primary transcripts (pri-miRNAs) (Kim 2005). Subsequently, the process to yield mature miRNAs involves two steps involving RNase-III enzymes and companion double-stranded RNA-binding domain (dsRBD) proteins (Figure 1).
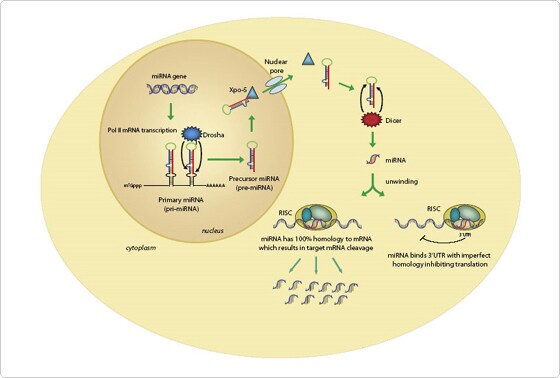
In the nucleus, the RNase III–type enzyme Drosha processes the long primary transcripts (pri-miRNA), yielding a hairpin precursors (pre-miRNA) consisting of approximately 70 nt. The pre-miRNA hairpins are exported to the cytoplasm where they are further processed into unstable, 19-25 nt miRNA duplex structures by the RNase III protein Dicer (Sontheimer 2005). The less stable of the two strands in the duplex is incorporated into a multiple-protein nuclease complex, the RNA-induced silencing complex (RISC), which regulates protein expression.
Mechanism of miRNA Gene Regulation
Plant miRNAs can bind to sequences on target mRNAs by exact or near-exact complementary base pairing and thereby direct cleavage and destruction of the mRNA (Rhoades 2002, Chen 2005) Similar to the mechanism employed in RNA interference (RNAi), the cleavage of a single phosphodiester bond on the target mRNA occurs between bases 10 and 11 (Elbashir et al. 2001). In contrast, nearly all animal miRNAs studied so far are usually not exactly complementary to their mRNA targets, and seem to inhibit protein synthesis while retaining the stability of the mRNA target (Ambros 2004). It has been suggested that transcripts may be regulated by multiple miRNAs and an individual miRNA may target numerous transcripts.
Additional Learning Topics
Interesting Review Articles
Bartel, D.P. MicroRNAs: Genomics, biogenesis, mechanism, and function. Cell. 2004; 116; 281-297.
Du, T. and Zamore, P.D. microPrimer: the Biogenesis and Function of microRNA. Development. 2005; 132: 4645-4652.
Pillai, R. S. MicroRNA Function: Multiple mechanisms for a tiny RNA? RNA. 2005; 11: 1753-1761.
Valencia-Sanchez, M.A., et.al. Control of Translation and mRNA Degradation by miRNAs and siRNAs. Genes & Development. 2006; 20: 515-524.
References Cited
Ambros, V. The Functions of Animal microRNAs Nature. 2004; 431: 350-355
Elbashir, S. et. al. RNA interference is mediated by 21 and 22 nt RNAs. Genes & Development. 2001; 15: 188-200.
Lagos-Quintana, M., et al. Identification of Novel Genes Coding for Small Expressed RNAs. Science. 2001;294: 853-858.
Lau, NC., et. al. An Abundant Class of Tiny RNAs with Probable Regulatory Roles in Caenorhabditis elegans. Science. 2001; 294: 858-862.
Lee, Y., et. al. MicroRNA Maturation: Stepwise Processing and Subcellular Localization. EMBO J.2002; 21: 4663-4670.
Lim LP., et. al. The microRNAs of Caenorhabditis elegans. Genes & Development. 2003; 17:991-1008.
Lim LP, et. al. Vertebrate microRNA Genes. Science. 2003; 299: 1540.
Rhoades MW., et. al. Prediction of plant microRNA targets. Cell. 2002; 110: 513-20.
Reinhart, BJ., et. al. The 21-nucleotide let-7 RNA Regulates Developmental Timing in Caenorhabditis elegans. Nature. 2000; 403: 901-906.
Rodriguez, A., et. al. Identification of Mammalian microRNA Host Genes and Transcription Units. Genome Research. 2004; 14: 1902-1910.
Sontheimer, E.J. Assembly and Function of RNA Silencing Complexes. Nature Reviews. Molecular Cellular Biology. 2005; 6: 127–138.
Literature
Questions
Send your questions to our top researchers at EpiScientist@thermofisher.com.