Search
Blocking Endogenous Targets Prior to IHC
Page contents
Endogenous biotin
Biotin is a vitamin and coenzyme in many biological reactions, and particularly those which take place in mitochondria. Also, biotin is conjugated to antibodies and enzymes for labeling and detection purposes because this permits the user to exploit the high affinity of biotin for special binding proteins like streptavidin, NeutrAvidin, and avidin. This is often referred to as the avidin-biotin complex or ABC.
Avidin-biotin complex methods are commonly used in IHC to enhance the detection of a target antigen. Because free or labeled strept (avidin) is used in these approaches, tissues rich in endogenous biotin, including liver, mammary gland, adipose tissue and kidney, can show high background staining. (Dakshinamurti, K. et al., 1963). Endogenous strept (avidin) binding activity due to high levels of endogenous biotin is usually most pronounced in frozen (cryostat) sections.
For any detection system where a biotin-binding protein is a component, endogenous biotin should be blocked to avoid recognition of endogenous biotin along with the target antigen. The basic procedure is as follows:
- Coat the sample with an excess of free, unlabeled strept(avidin).
- Add an excess of free biotin to fill all unoccupied biotin-binding sites in the tetrameric strept(avidin) molecules (Wood, G.S. et al., 1981)
The end result is that all endogenous biotin is bound to strept(avidin), and all unoccupied biotin-binding sites on each strept(avidin) molecule are filled. Because heat-induced epitope retrieval (HIER) may increase the level of endogenous biotin that becomes detectable in a sample, negative control samples should also undergo HIER to reduce the possibility of false positives from increased biotin detection.
Egg white avidin is highly glycosylated, and these polysaccharides can bind to sugar-binding lectins in tissues and increase the background signal. One early approach to preventing this interaction is to use an analog of the avidin glycosylation to saturate these binding sites on the tissue sample (Naritoku, W.Y. et al., 1982). More recently, avidin has been replaced almost totally with its non-glycosylated homologue streptavidin or NeutrAvidin , a de-glycosylated form of avidin, for eliminating endogenous biotin. This representative immunofluorescence analysis in A549 cells provides an example of how the use of endogenous biotin-blocking reagents may be used to reduce signal: noise ratio.
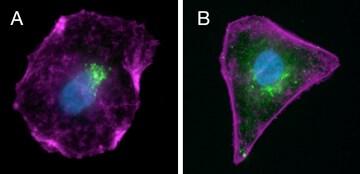
Reducing endogenous background. A549 cells were fixed and permeabilized, treated (A) or not treated (B) with the Endogenous Biotin–Blocking Kit (E21390), labeled with a primary antibody against mouse golgin-97 followed by DSB-X biotin–conjugated goat anti–mouse IgG, then stained with Alexa Fluor 488 streptavidin (green), Alexa Fluor 647 phalloidin (magenta), and NucBlue Fixed Cell ReadyProbes Reagent (blue). Note the green fluorescence produced by the labeled streptavidin is punctate and limited to the target Golgi apparatus in the treated cell (A), but there is considerable diffuse, nonspecific green fluorescence in the untreated cell (B), in addition to the punctate staining. The image was acquired at 60x magnification.
Learn more
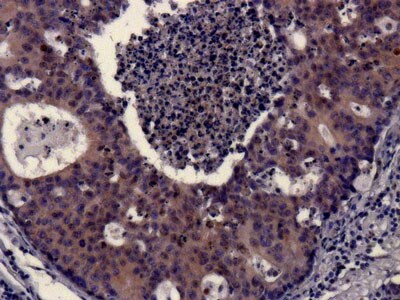
Chromogenic IHC detection of cytokeratin 18 in colon carcinoma tissue. Endogenous peroxidase was quenched with Thermo Fisher Peroxidase Suppressor (35000) and endogenous biotin was blocked prior to incubating the sample with a biotinylated cytokeratin 18 antibody (e.g. MA512101). Cytokeratin 18 was detected with Thermo Fisher NeutrAvidin High Sensitivity HRP Conjugate (31032) in combination with Thermo Fisher Metal-Enhanced DAB (34065). Cell nuclei were counterstained with hematoxylin (e.g. TA-060-MH).
Peroxidases, most often horseradish peroxidase (HRP), are commonly used as the enzyme catalyst for chromogenic substrate-based staining of target antigens in IHC. Imaging this type of IHC result can be complicated by the presence of endogenous peroxidase and "pseudoperoxidase" activity in the cells and tissues. Endogenous peroxidase reacts with hydrogen peroxide to reduce the 3, 3’-diaminobenzidine (DAB) substrate or other peroxidase substrates, resulting in nonspecific staining of the tissue.
To confirm the presence of endogenous peroxidase activity, the re-hydrated tissue sections should be reacted with a peroxidase substrate such as DAB Any colored precipitate that forms in the tissue from this treatment indicates endogenous activity. If a tissue is rich in endogenous peroxidase activity that cannot be eliminated, an alternative enzyme label such as alkaline phosphatase (AP) can be used instead of HRP
Several methods have been devised to inhibit or destroy endogenous peroxidase activity after tissue fixation. The removal of endogenous peroxidases can be performed before or after antigen retrieval or before and after the primary antibody incubation step. However, it may be difficult to suppress all endogenous peroxidase activity in tissues containing large numbers of erythrocytes (RBC). Also note that if you are staining surface antigens on cells like CD4 and CD8, blocking endogenous peroxidase activity should be done after the primary or secondary antibody incubation step. Otherwise, the blocking step will destroy these antigens.
Typical methods for quenching endogenous peroxidase activity in tissue sections are as follows: Prepare the peroxidase blocking solution by adding one part 30% hydrogen peroxide to nine parts methanol. Submerge the slides in this solution for 10 min. Wash three times with PBS, and then proceed with the rest of the staining protocol. If a 3% (v/v/) final concentration of hydrogen peroxide damages your tissue sections and/or alters epitope accessibility, try reducing the concentration to 0.3% (v/v).
Our product Hydrogen Peroxide Block (TA-060-HP, TA-125-HP) is recommended for use after the de-paraffinization and re-hydration steps. The tissue sections are incubated in this blocker for 10-15 min at ambient temperature or at 37°C, and then washed twice with buffer prior to continuing the staining protocol. We have another pre-made peroxidase blocker called Peroxidase Suppressor (35000). An illustration of its effects are shown below.
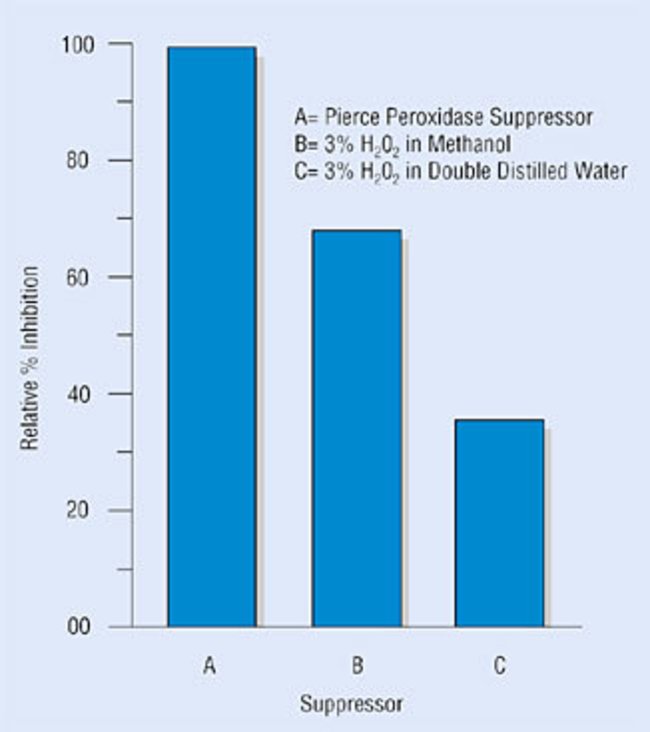
Endogenous peroxidase activity in frozen (cryo) sections. Thermo Scientific Peroxidase Suppressor was used to inhibit endogenous peroxidase activity by incubating tissue sections or cells for 10-15 min in a solution of 0.3% (v/v) hydrogen peroxide in aqueous 0.1% (w/v) sodium azide. This process reduces background signal in HRP-based immunohistochemistry applications.
Learn more
Phosphatases hydrolyze phosphate groups on cellular substrates, and alkaline phosphatase (AP), usually isolated from calf intestine, and is commonly used as a reporter for IHC detection. Endogenous phosphatases can react with the AP substrates used for IHC, such as a combination of nitro blue tetrazolium chloride (NBT) and 5-bromo-4-chloro-3-indolyl phosphate (BCIP), resulting in nonspecific staining.
A similar test, as described above for peroxidases, can be employed using NBT/BCIP to determine if endogenous phosphatases need to be quenched. If they do, then the AP inhibitor levamisole can be added at a final concentration of 1 mM between the primary and secondary antibody incubation steps to inhibit endogenous phosphatase activity. An NBT/BCIP/levamisole mixture designed for this purpose is product 34042. Also, endogenous phosphatase activity is easily destroyed by boiling the sections, as in the typical HIER procedure.
- Dakshinamurti, K. and Mistry, S.P. (1963) J. Biol. Chem. 238, 294.
- Naritoku, W.Y. and Taylor, C.R. (1982) J. Histochem. Cytochem. 30, 253-260.
- Coons, A.A., et al. (1942) J. Immunol. 45, 159-170
- Beisker W et al. (1987) Cytometry 8:235–239.
- Cowen T et al. (1985) Histochemistry 82:205–208.
- Mosiman VL et al. (1997) Cytometry 30:151–156.
- Romijn, Herms J. et al. (1999) J Histochem Cytochem 47:229–236.
For Research Use Only. Not for use in diagnostic procedures.