Search
Primer Designer Tool FAQs
Below are frequently asked questions when using our online Primer Designer™ Tool to search for the right PCR/Sanger primer pair from a database of ~650,000 pre-designed primer pairs for re-sequencing the human exome and human mitochondrial genome.
Q. What is the delivery time frame for predesigned primer pairs?
A. Typical delivery times for USA and Canada are listed below:
a. Desalted primers: delivery within 3 business days
b. HLPC-purified primers: delivery within 8 business days
Q: How do I know which area is sequenced?
A: For each amplicon, the amplicon coordinates are displayed. Clicking on “View Primer on Map” displays the amplicons on the genome and in the context of transcripts.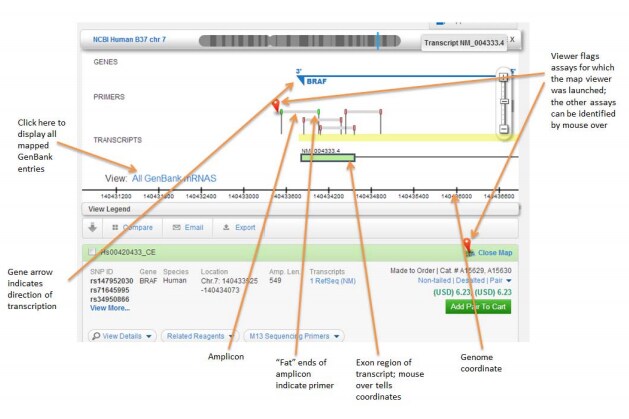
Q: What are the supported search terms?
A: Assay ID: e.g. "Hs00326060_CE" SNP ID: e.g. "rs12096805" Entrez Gene ID: e.g. "6262 Gene Symbol: e.g. "JAK2" Gene Name: e.g. "Janus kinase 2" RefSeq Accession #: e.g. "NM_007294" GenBank mRNA: e.g. CR536579.1" Protein ID: e.g. "CAA66975.1" Cosmic ID: e.g. "121007" The Primer Designer™ Tool also allows one to enter a sequence and mapping primer pairs will be returned.
Q: How do I find primers for a specific exon?
A: Exon counting is not standardized. Exon 1 often means the first exon on the transcript, but could also mean the first exon on the genome regardless the sense of transcription. The mouse over pop‐ up‐box on the genomic viewer indicates the number of an exon. However, we recommend confirmation by the customer to ensure that the exon number indicated is concordant with the exon counting system the user expects.
Q: What should the distance from a SNP of interest be relative to the 3’ end of the primer?
A: Depending on the sequencing chemistry, we recommend choosing primer pairs that will result in amplicons where the variant of interest is located away from the 3' end of the sequencing primer. BigDye™ Direct and BigDye™ Terminator v1.1 typically have higher data quality very close to the sequencing primer end, whereas BigDye Terminator v3.1 together with POP‐7™ polymer provides optimized data quality after the first 35 to 40 bases.
Q: Are primers in exon regions?
A: The primer pairs are amplifying exon regions of genes. In order to have complete coverage the first and last primer of each exon lies in an intron.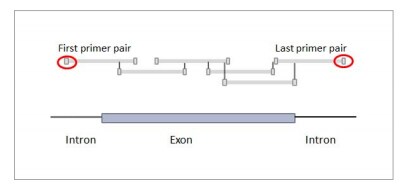
Q: How large is the intron span on either end?
A: Currently, we provide only primers for exons and are not offering “intron only” amplicons. Many of the primer pairs span the entire exon, and hence provide some sequence for exon adjacent sequence. The extent of this intronic sequence varies from assay to assay and the extent can be viewed in the genome browser panel.
Q: How can I obtain the amplicon sequence for a primer pair?
A: The amplicon coordinates are displayed for each primer pair. At this time we do not provide the amplicon sequences from our webpage. To obtain the amplicon sequence, we recommend the NCBI website. You can click on View Details within the Primer Designer Tool and there are several hyperlinks that will take you to different locations on the NCBI page. For example, if you click on the Entrez Gene ID hyperlink, it will take you to the Gene ID page. You can scroll down to Genomic regions, transcripts, and products and click on the FASTA option next to "Go to nucleotide" and that will bring up the whole sequence.
Q: Who can I contact if I do not receive my primers after ordering?
A: Contact Thermo Fisher Scientific Product Support and have your order number handy.
Q: What should I do if my primers do not work due to no amplification, off‐target amplification or SNP of interest too close to the PCR priming region?
A: Contact Thermo Fisher Product Support.
Product literature
For Research Use Only. Not for use in diagnostic procedures.