Search
Real-time PCR Goes Prime Time
Real-time PCR assays used for quantitative RT-PCR combine the best attributes of both relative and competitive (end-point) RT-PCR in that they are accurate, precise, capable of high throughput, and relatively easy to perform.
To truly appreciate the benefits of this technology, a review of PCR fundamentals is necessary. At the start of a PCR reaction, reagents are in excess, template and product are at low enough concentrations that product renaturation does not compete with primer binding, and amplification proceeds at a constant, exponential rate. Exactly when the reaction rate ceases to be exponential and enters a linear phase of amplification is extremely variable, even among replicate samples, but it appears to be primarily due to product renaturation competing with primer binding (since adding more reagents or enzyme has little effect). At some later cycle the amplification rate drops to near zero (plateaus), and little more product is made.
For the sake of accuracy and precision, it is necessary to collect quantitative data at a point in which every sample is in the exponential phase of amplification (since it is only in this phase that amplification is extremely reproducible). Analysis of reactions during exponential phase at a given cycle number should theoretically provide several orders of magnitude of dynamic range. Rare targets will probably be below the limit of detection, while abundant targets will be past the exponential phase. In practice, a dynamic range of 2-3 logs can be quantitated during end-point relative RT-PCR. In order to extend this range, replicate reactions may be performed for a greater or lesser number of cycles, so that all of the samples can be analyzed in the exponential phase.
Real-time PCR automates this otherwise laborious process by quantitating reaction products for each sample in every cycle. The result is an amazingly broad 107-fold dynamic range, with no user intervention or replicates required. Data analysis, including standard curve generation and copy number calculation, is performed automatically. As more labs and core facilities acquire the instrumentation required for real-time analysis, this technique may become the dominant RT-PCR-based quantitation technique.
To truly appreciate the benefits of this technology, a review of PCR fundamentals is necessary. At the start of a PCR reaction, reagents are in excess, template and product are at low enough concentrations that product renaturation does not compete with primer binding, and amplification proceeds at a constant, exponential rate. Exactly when the reaction rate ceases to be exponential and enters a linear phase of amplification is extremely variable, even among replicate samples, but it appears to be primarily due to product renaturation competing with primer binding (since adding more reagents or enzyme has little effect). At some later cycle the amplification rate drops to near zero (plateaus), and little more product is made.
For the sake of accuracy and precision, it is necessary to collect quantitative data at a point in which every sample is in the exponential phase of amplification (since it is only in this phase that amplification is extremely reproducible). Analysis of reactions during exponential phase at a given cycle number should theoretically provide several orders of magnitude of dynamic range. Rare targets will probably be below the limit of detection, while abundant targets will be past the exponential phase. In practice, a dynamic range of 2-3 logs can be quantitated during end-point relative RT-PCR. In order to extend this range, replicate reactions may be performed for a greater or lesser number of cycles, so that all of the samples can be analyzed in the exponential phase.
Real-time PCR automates this otherwise laborious process by quantitating reaction products for each sample in every cycle. The result is an amazingly broad 107-fold dynamic range, with no user intervention or replicates required. Data analysis, including standard curve generation and copy number calculation, is performed automatically. As more labs and core facilities acquire the instrumentation required for real-time analysis, this technique may become the dominant RT-PCR-based quantitation technique.
Real-time Reporters: SYBR® Green, TaqMan®, and Molecular Beacons
All real-time PCR systems rely upon the detection and quantitation of a fluorescent reporter, the signal of which increases in direct proportion to the amount of PCR product in a reaction. In the simplest and most economical format, that reporter is the double-strand DNA-specific dye SYBR® Green (Molecular Probes). SYBR Green binds double-stranded DNA, and upon excitation emits light. Thus, as a PCR product accumulates, fluorescence increases.
The advantages of SYBR Green are that it's inexpensive, easy to use, and sensitive. The disadvantage is that SYBR Green will bind to any double-stranded DNA in the reaction, including primer-dimers and other non-specific reaction products, which results in an overestimation of the target concentration. For single PCR product reactions with well designed primers, SYBR Green can work extremely well, with spurious non-specific background only showing up in very late cycles.
The two most popular alternatives to SYBR Green are TaqMan® and molecular beacons, both of which are hybridization probes relying on fluorescence resonance energy transfer (FRET) for quantitation.
TaqMan Probes are oligonucleotides that contain a fluorescent dye, typically on the 5' base, and a quenching dye, typically located on the 3' base. When irradiated, the excited fluorescent dye transfers energy to the nearby quenching dye molecule rather than fluorescing, resulting in a nonfluorescent substrate. TaqMan probes are designed to hybridize to an internal region of a PCR product. During PCR, when the polymerase replicates a template on which a TaqMan probe is bound, the 5' exonuclease activity of the polymerase cleaves the probe. This separates the fluorescent and quenching dyes and FRET no longer occurs. Fluorescence increases in each cycle, proportional to the rate of probe cleavage.
Molecular beacons also contain fluorescent and quenching dyes, but FRET only occurs when the quenching dye is directly adjacent to the fluorescent dye. Molecular beacons are designed to adopt a hairpin structure while free in solution, bringing the fluorescent dye and quencher in close proximity. When a molecular beacon hybridizes to a target, the fluorescent dye and quencher are separated, FRET does not occur, and the fluorescent dye emits light upon irradiation. Unlike TaqMan probes, molecular beacons are designed to remain intact during the amplification reaction, and must rebind to target in every cycle for signal measurement.
The advantages of SYBR Green are that it's inexpensive, easy to use, and sensitive. The disadvantage is that SYBR Green will bind to any double-stranded DNA in the reaction, including primer-dimers and other non-specific reaction products, which results in an overestimation of the target concentration. For single PCR product reactions with well designed primers, SYBR Green can work extremely well, with spurious non-specific background only showing up in very late cycles.
The two most popular alternatives to SYBR Green are TaqMan® and molecular beacons, both of which are hybridization probes relying on fluorescence resonance energy transfer (FRET) for quantitation.
TaqMan Probes are oligonucleotides that contain a fluorescent dye, typically on the 5' base, and a quenching dye, typically located on the 3' base. When irradiated, the excited fluorescent dye transfers energy to the nearby quenching dye molecule rather than fluorescing, resulting in a nonfluorescent substrate. TaqMan probes are designed to hybridize to an internal region of a PCR product. During PCR, when the polymerase replicates a template on which a TaqMan probe is bound, the 5' exonuclease activity of the polymerase cleaves the probe. This separates the fluorescent and quenching dyes and FRET no longer occurs. Fluorescence increases in each cycle, proportional to the rate of probe cleavage.
Molecular beacons also contain fluorescent and quenching dyes, but FRET only occurs when the quenching dye is directly adjacent to the fluorescent dye. Molecular beacons are designed to adopt a hairpin structure while free in solution, bringing the fluorescent dye and quencher in close proximity. When a molecular beacon hybridizes to a target, the fluorescent dye and quencher are separated, FRET does not occur, and the fluorescent dye emits light upon irradiation. Unlike TaqMan probes, molecular beacons are designed to remain intact during the amplification reaction, and must rebind to target in every cycle for signal measurement.
Real-time Reporters for Multiplex PCR
TaqMan probes and molecular beacons allow multiple DNA species to be measured in the same sample (multiplex PCR), since fluorescent dyes with different emission spectra may be attached to the different probes. Multiplex PCR allows internal controls to be co-amplified and permits allele discrimination in single-tube, homogeneous assays. These hybridization probes afford a level of discrimination impossible to obtain with SYBR Green, since they will only hybridize to true targets in a PCR and not to primer-dimers or other spurious products.
Taqman vs. SYBR Green: The Data
An example of the precision and accuracy of real-time RT-PCR is shown in Figure 1. Panels A and B (left) are the amplification profiles of serial 107-fold dilutions of a cDNA synthesis reaction (107 fold range), amplified with Ambion's QuantumRNA™ 18S Universal primers and detected by either SYBR Green (panel A) or TaqMan (panel B) chemistries. Standard curves for both are shown on the right. The only practical difference between the performance of these two methods is the occasional presence of a false signal late in the SYBR Green amplification. This usually corresponds to a signal lower than that expected from femtograms of RNA, and is of little consequence to accuracy except if a target RNA is extremely rare.
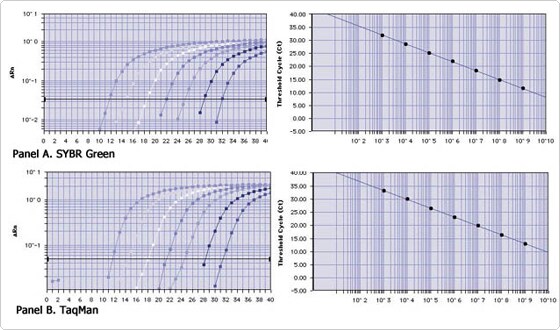
Figure 1. Precision and Accuacy of Real-Time RT-PCR. 18S rRNA amplification profiles and standard curves generated using A) SYBR Green, and B) TaqMan chemistries.

Figure 1. Precision and Accuacy of Real-Time RT-PCR. 18S rRNA amplification profiles and standard curves generated using A) SYBR Green, and B) TaqMan chemistries.
Investing in the Real-Time Technique
Real-time PCR requires an instrumentation platform that consists of a thermal cycler, computer, optics for fluorescence excitation and emission collection, and data acquisition and analysis software. These machines, available from several manufacturers, differ in sample capacity (some are 96-well standard format, others process fewer samples or require specialized glass capillary tubes), method of excitation (some use lasers, others broad spectrum light sources with tunable filters), and overall sensitivity. There are also platform-specific differences in how the software processes data. Real-time PCR machines are not cheap, currently about $60-$95K, but are well within purchasing reach of core facilities or labs that have the need for high throughput quantitative analysis.
Supporting Products
Ambion has many products useful for real-time RT-PCR and that are routinely used for such analysis at Ambion in the ABI PRISM® 7700 Sequence Detection System.
Ambion's RETROscript™ Kit is a versatile and robust reverse transcription kit containing M-MLV enzyme, buffers and dNTPs for reverse transcription, and both oligo(dT) and random primers. A control RNA and primers are also supplied with the kit. SuperTaq™ DNA Polymerase is a thermostable polymerase for amplification of products up to 1 kb. For longer amplification products, SuperTaq Plus™ is recommended. Both are supplied with a buffer with and without MgCl2 and a separate tube of MgCl2 for Mg2+ optimization, as well as dNTPs.
RNAqueous™-4PCR and RNAqueous™-96 are RNA isolation kits specifically designed for high throughput of small samples for RT-PCR applications. Both contain Ambion's RNase-free DNase I. These products are described in more detail in " RNA Isolation for RT-PCR". DNA-free™ can be used to remove contaminating DNA from RNA preparations made using your own isolation protocol, without phenol extraction, heating or precipitation. 96-well PCR plates and optically clear strip caps are also available.
All of the above mentioned products are compatible with SYBR Green, TaqMan, and molecular beacon chemistries. In addition, the QuantumRNA 18S Universal and ß-actin standards work well with SYBR Green chemistry, and a QuantumRNA 18S universal TaqMan probe sequence is available from Technical Service upon request.
Ambion's RETROscript™ Kit is a versatile and robust reverse transcription kit containing M-MLV enzyme, buffers and dNTPs for reverse transcription, and both oligo(dT) and random primers. A control RNA and primers are also supplied with the kit. SuperTaq™ DNA Polymerase is a thermostable polymerase for amplification of products up to 1 kb. For longer amplification products, SuperTaq Plus™ is recommended. Both are supplied with a buffer with and without MgCl2 and a separate tube of MgCl2 for Mg2+ optimization, as well as dNTPs.
RNAqueous™-4PCR and RNAqueous™-96 are RNA isolation kits specifically designed for high throughput of small samples for RT-PCR applications. Both contain Ambion's RNase-free DNase I. These products are described in more detail in " RNA Isolation for RT-PCR". DNA-free™ can be used to remove contaminating DNA from RNA preparations made using your own isolation protocol, without phenol extraction, heating or precipitation. 96-well PCR plates and optically clear strip caps are also available.
All of the above mentioned products are compatible with SYBR Green, TaqMan, and molecular beacon chemistries. In addition, the QuantumRNA 18S Universal and ß-actin standards work well with SYBR Green chemistry, and a QuantumRNA 18S universal TaqMan probe sequence is available from Technical Service upon request.