Search
RNAi and Non-coding RNA Research
RNA interference (RNAi) is a very powerful tool for studying the basic biology of cells, allowing the knockdown of gene expression to study protein function in a wide range of cell types. Once viewed as a technique used only by select laboratories, RNAi is now considered essential for studying gene function. It has become a prominent tool for protein knockdown studies, phenotype analysis, function recovery, pathway analysis, in vivo knockdown, and drug target discovery.
Glossary of common RNAi terms
Ribonucleic acid interference (first used by A. Fire and C. Mello et al., 1998).
Short interfering RNA. siRNAs are 21–25 bp dsRNAs with dinucleotide 3’ overhangs and are processed from longer dsRNA by Dicer in the RNA interference pathway. Introduction of synthetic siRNAs can induce RNAi in mammalian cells. siRNAs can also originate from endogenous precursors.
Short hairpin RNA; also short interfering hairpin. shRNAs are used in vector-based approaches for supplying siRNA to cells for stable gene silencing. A strong Pol III-type promoter is used to drive transcription of a target sequence designed to form hairpins and loops of variable length, which are processed by cellular siRNA machinery. Once in the cell, the shRNA can decrease the expression of a gene with complementary sequences by RNAi.
Vectors that express microRNAs for RNAi. miRNAs are 19–23 nt single-stranded RNAs, originating from single-stranded precursor transcripts that are characterized by imperfectly base-paired hairpins. miRNAs function in a silencing complex that is similar, if not identical, to RISC (see below).
siRNA molecules which have chemical modifications.
RNA-induced silencing complex (RISC). A nuclease complex composed of proteins and siRNA that targets and cleaves endogenous mRNAs complementary to the siRNA within the RISC complex.
Effects that occur when one or a few genes not specifically targeted show loss of gene function following the introduction of an siRNA or d-siRNA pool. The effect may be mediated by the sense strand of an siRNA, which may initiate a loss-of-function response from an unrelated gene. Off-target effects can also occur as a secondary effect of the antisense strand of a specific siRNA, if it has sufficient homology to knock down the expression of a non-target gene.
How RNAi works
Two types of small RNA molecules function in RNAi. The first are synthetic, short interfering RNA (siRNA) molecules that target mRNA cleavage, effectively knocking down the expression of a gene of interest. MicroRNA (miRNA) molecules, on the other hand, are naturally occurring single-stranded RNAs 19–22 nucleotides long, which regulate gene expression by binding to the 3’ untranslated regions (UTRs) of target mRNAs and inhibiting their translation (Ambros, 2004).
- Learn more about RNAi
siRNA analysis
There are several ways to induce RNAi: synthetic molecules, RNAi vectors, and in vitro dicing (Figure 1, below). In mammalian cells, short pieces of dsRNA—short interfering RNA— initiate the specific degradation of a targeted cellular mRNA. In this process, the antisense strand of siRNA becomes part of a multiprotein complex, or RNA-induced silencing complex (RISC), which then identifies the corresponding mRNA and cleaves it at a specific site. Next, this cleaved message is targeted for degradation, which ultimately results in the loss of protein expression.
- Learn more about siRNA analysis
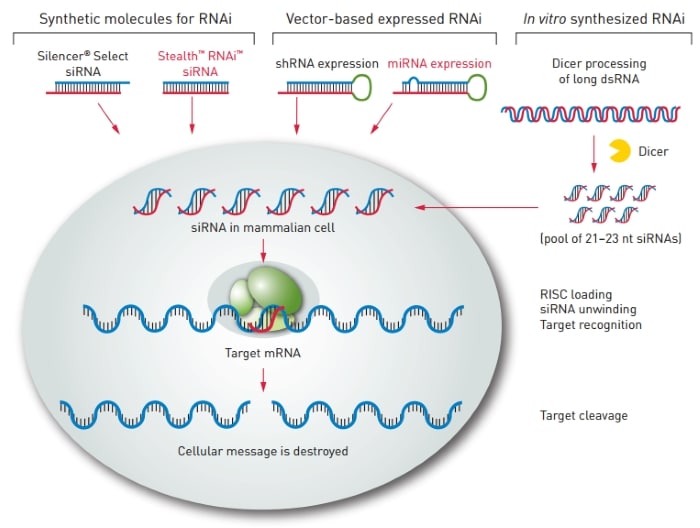
Figure 1: Methods of RNAi knockdown in mammalian cells.
miRNA analysis
Both RNA polymerase II and III transcribe miRNA-containing genes, generating long primary transcripts (pri-miRNAs) that are processed by the RNase III–type enzyme Drosha, yielding hairpin structures 70 to 90 bp in length (pre-miRNAs). Pre-miRNA hairpins are exported to the cytoplasm, where they are further processed by the RNase III protein Dicer into short double-stranded miRNA duplexes 19 to 22 nucleotides long. The miRNA duplex is recognized by the RNA-induced silencing complex (RISC), a multiple protein nuclease complex, and one of the two strands, the guide strand, assists this protein complex in recognizing its cognate mRNA transcript. The RISC-miRNA complex often interacts with the 3’ UTR of target mRNAs at regions exhibiting imperfect sequence homology, inhibiting protein synthesis by a mechanism that has yet to be fully elucidated (Figure 2, below).
Plant miRNAs can bind to sequences on target mRNAs by exact or near-exact complementary base pairing and thereby direct cleavage and destruction of the mRNA (Rhoades et al., 2002; Chen, 2005). Similar to the mechanism employed in RNA interference (RNAi), the cleavage of a single phosphodiester bond on the target mRNA occurs between bases 10 and 11 (Elbashir et al., 2001). In contrast, nearly all animal miRNAs studied so far do not exhibit perfect complementarity to their mRNA targets, and seem to inhibit protein synthesis while retaining the stability of the mRNA target (Ambros, 2004). It has been suggested that transcripts may be regulated by multiple miRNAs, and an individual miRNA may target numerous transcripts. Research suggests that as many as one-third of human genes may be regulated by miRNAs (Lim et al., 2003). Although hundreds of miRNAs have been discovered in a variety of organisms, little is known about their cellular function. Several unique physical attributes of miRNAs, including their small size, lack of polyadenylated tails, and tendency to bind their mRNA targets with imperfect sequence homology, have made them elusive and challenging to study.
- Learn more about miRNA analysis
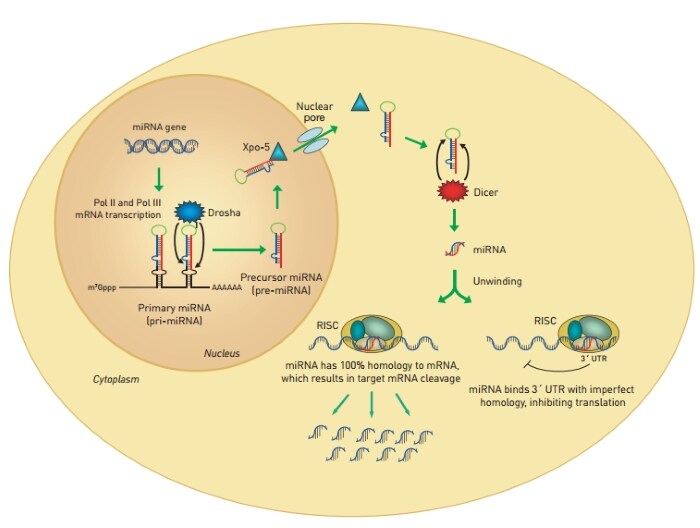
Figure 2: Biogenesis and function of miRNA. MicroRNA transcripts, generated by RNA polymerases II and III, are processed by the RNase III enzymes Drosha (nuclear) and Dicer (cytoplasmic), yielding 19–22 nucleotide miRNA duplexes. One of the two strands of the duplex is incorporated into the RISC complex, which regulates protein expression.
Choosing and RNAi approach
The process of RNAi (RNA interference) can be moderated by either siRNA or miRNA. Both are processed inside the cell by the enzyme called Dicer and incorporated into a complex called RISC (RNA-induced silencing complex). However, there are subtle differences between the two.
siRNA is an exogenous double-stranded RNA that can either be chemically synthesized and then directly transfected into cells, or generated inside the cell by introducing vectors that express short-hairpin RNA (shRNA), which are the precursors of siRNAs. miRNA, on the other hand, is single stranded and comes from endogenous non-coding RNA found within the introns of larger RNA molecules. However, the processing of shRNA into functional siRNA involves the same cellular RNAi machinery that naturally processes genome-encoded miRNAs, which are responsible for cellular regulation of gene expression by modulating mRNA stability, translation, and chromatin structures (Hutvagner and Zamore, 2002).
Another difference between siRNA and miRNA is that siRNA typically binds perfectly and specifically to its mRNA target in animals, while miRNA can inhibit translation of many different mRNA sequences because its pairing is imperfect. In plants, miRNA tends to have a more perfectly complimentary sequence, which induces mRNA cleavage as opposed to just repression of translation.
Both siRNA and miRNA can play a role in epigenetics through a process called RNA induced transcriptional silencing (RITS). Likewise, both are important targets for therapeutic use because of the roles they play in the controlling gene expression.
| siRNA | miRNA | |
|---|---|---|
| Occurrence | Occurs naturally in plants and lower animals. Whether or not they occur naturally in mammals is an unsettled question. | Occurs naturally in plants and animals. |
| Configuration | Double stranded | Single stranded |
| Length | 21-22 nt | 19-25 nt |
| Complementarity to target mRNA | 100% perfect match; therefore, siRNAs knock down specific genes, with minor off-target exceptions. | Not exact; therefore, a single miRNA may target up to hundreds of mRNAs. |
| Biogenesis | Regulate the same genes that express them. | Expressed by genes whose purpose is to make miRNAs, but they regulate genes (mRNAs) other than the ones that expressed them. |
| Action | Cleave mRNA | Inhibit translation of mRNA |
| Function | Act as gene silencing guardians in plants and animals that do not have antibody-or cell-mediated immunity. | Regulators (inhibitors) of genes (mRNAs) |
| Uses | siRNAs are valuable laboratory tools used in nearly every molecular biology laboratory to knock down genes. Several siRNAs are in clinical trials as possible therapeutic agents. | Possible therapeutic uses either as drug targets or as drug agents themselves. Expression levels of miRNAs can be used as potential diagnostic and biomarker tools. |
* Table adapted from Mack, 2007
For Research Use Only. Not for use in diagnostic procedures.