Search
Identification of Putative Limbal Stem Cells in Populations of Normal Human Corneal Epithelial Cells
It is widely accepted that the stem cells which maintain and renew the corneal epithelium reside in the limbus, located on the periphery of the cornea (Figure 1, A and B). They are commonly referred to as limbal stem cells (LSCs) and are useful for basic research into various aspects of corneal biology such as ocular injuries that impair vision [1,2]. Progress in this research has been hampered, however, by the lack of markers and assays that can be used to identify LSCs. Here we present a method for identification and quantification of LSCs derived from primary human tissue, using the Attune® Acoustic Focusing Cytometer.
Side Population Analysis Facilitated With the Attune® Cytometer
Many types of stem cells can be identified using a technique known as side population (SP) analysis [3]. This approach takes advantage of stem cells’ relatively high expression of membrane transporter proteins, such as ABCG2, that can specifically efflux various compounds, including fluorescent dyes, from live cells. Once stem cells begin to differentiate, expression of these transporters is reduced and cells retain the dye molecules. We adapted the SP analysis technique for identification of limbal stem cells in populations of human corneal epithelial cells. To do this we took advantage of the ability of Vybrant® DyeCycle™ Violet stain to be effluxed by the ABCG2 transporter. We labeled cells with this cell-permeant, fluorescent DNA-binding dye, then analyzed the population using the rare-event detection capability of the Attune® Acoustic Focusing Cytometer.
- Human corneal epithelial cells (HCECs) were isolated via enzymatic digestion of limbal sections from which the central cornea and most of the sclera were trimmed (Figure 1, A and B).
- Epithelial cells released from these sections were subsequently expanded through two passages in Gibco® Keratinocyte-SFM, then cryopreserved.
- HCECs were then thawed and passaged twice in Keratinocyte-SFM. Subconfluent cells (Figure 1C) were detached from the culture dish, resuspended at 1 x 106 cells/mL in culture medium and incubated with 5 µM Vybrant® DyeCycle™ Violet stain for 60 min at 37°C with or without the ABCG2 inhibitor fumitremorgin C (1 µM).
- Cells were subsequently washed with cold HBSS with 2% FBS and resususpended in culture media with 5 µM Vybrant® DyeCycle™ Violet stain with or without 1 µM fumitremorgin C for an additional 30 min at 37°C.
- Cells were washed once and resuspended in culture medium prior to analysis.
The Attune® Acoustic Focusing Cytometer was used to analyze ~120,000 cells per experimental replicate. Vybrant® DyeCycle™ Violet stain was excited using the 405 nm laser and emission was collected in 450/40 and 603/48 bandpass filters.
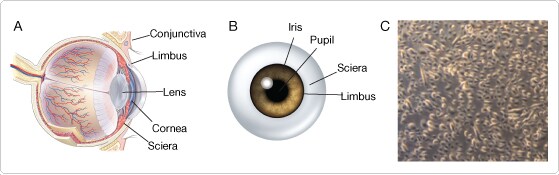
Figure 1. Diagram of the human cornea and photomicrograph of isolated HCECs. (A) Lateral and (B) Anterior illustrations depicting anatomy of the human eye. Note that the limbus is localized between the iris and conjunctival tissue of the sclera. (C) Phase-contrast photomicrograph of subconfluent HCECs cultured in Keratinocyte-SFM.
Results
Dual-emission fluorescence analysis using acoustic focusing cytometry provided clear identification of the side population of limbal stem cells (Figure 2). Results were supported by the inhibition of ABCG2-mediated dye efflux using the inhibitor fumitremorgin C (Figure 2B). With its superior cell focusing and high sample collection rates, the Attune® Acoustic Focusing Cytometer facilitated the analysis of large numbers of putative stem cells in minimal time, while maintaining high data integrity.
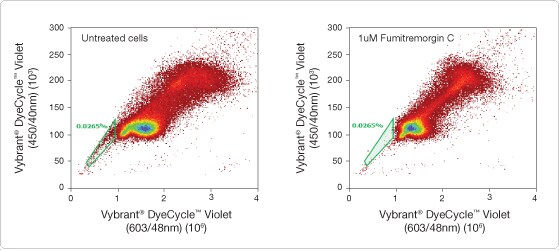
Figure 2. Side population analysis of HCECs. (Left) In untreated cells, a side population of 0.452% (green-gated area) of cells excluded Vybrant® DyeCycle™ Violet stain; this population represents putative LSCs. (Right) Cells treated with 1 µM fumitremorgin C, an ABCG2 inihitor, showed a reduction in the HCEC side population to 0.0265% (green-gated area). Note that similar reductions in side population counts were also observed when cells were co-incubated with higher levels of fumitremorgin C (5 and 10 µM, data not shown).
References
- Rama P, Matuska S, Paganoni G et al. (2010) Limbal stem-cell therapy and long-term corneal regeneration. N Engl J Med 363(2):147–155. Epub 2010 Jun 23.
- Baylis O, Figueiredo F, Henein C et al. (2011) 13 years of cultured limbal epithelial cell therapy: a review of the outcomes. J Cell Biochem 112(4):993–1002. doi: 10.1002/jcb.23028.
- Telford WG, Bradford J, Godfrey W et al. (2007) Side population analysis using a violet-excited cell-permeable DNA binding dye. Stem Cells 25(4):1029–1036. Epub 2006 Dec 21.
For Research Use Only. Not intended for any animal or human therapeutic or diagnostic use.
For Research Use Only. Not for use in diagnostic procedures.
Get a copy of this article as it appears in the print version of BioProbes 66.
See a complete listing of the products discussed in this article.
View Products
