Search
Simplify Sample Preparation Without Compromising Data Using the Attune
Immunophenotyping whole blood using traditional flow cytometry is typically performed with lysis of red blood cells (RBCs), and protocols most often include a wash step (lyse + wash). RBC lysis and wash steps can result in significant cell loss and damage [1]. While a no-lyse, no-wash method for immunophenotyping of whole blood has been previously described, the method is of limited utility in traditional systems, which use hydrodynamic forces to orient cells in the flow stream [2]. Moreover, significant sample dilution is required in no-lyse, no-wash methods to achieve low coincidence with red blood cells, necessitating extremely long acquisition times. Immunophenotyping with samples of limited volume, such as mouse whole blood (≤100 μL/day/animal) [3,4], is particularly challenging—these small volumes are generally not sufficient for multicolor phenotyping experiments with the required compensation and fluorescence-minus-one (FMO) controls [5].With the Attune® cytometer, you can acquire data quickly, even from dilute samples, without compromising data (Figure 1). Difficult-to-collect samples like mouse blood and bone marrow, thin-needle aspirates, or any sample with low cell yield can be stained and then diluted without washing or performing RBC lysis. High-rate collection makes acquiring these types of samples possible—you can run up to 4 mL typically in just 4 minutes. And because the preparation steps that often lead to sample loss are eliminated, full-panel testing is possible even for precious samples.
 | Figure 1. Acoustic focusing compared to traditional hydrodynamic focusing. (A) In acoustic focusing, cells remain in tight alignment even at higher sample rates. With this tight alignment, cells pass through the laser beam at its optimal focal point, resulting in less signal variation and improved data quality. (B) In traditional hydrodynamic focusing, increasing the sample rate results in widening of the sample core stream. The speed at which cells pass through the laser is not changed, and is determined by the speed of the sheath fluid flow. Cells are distributed throughout the sample core stream because of reduced differential pressure between sample stream and sheath stream, resulting in reduced cell focusing. Cells are not in tight alignment as they pass through the laser beam, resulting in increased signal variation and compromised data quality. |
Minimizing Sample Preparation Without Compromising Data
The Attune® Acoustic Focusing Cytometer aligns cells using acoustic forces independent of the fluid stream. This precise alignment delivers much higher throughput than is possible with traditional flow cytometry (Figure 1). With significantly higher sample throughput (as much as 10 times faster than other cytometers and up to 1 mL per minute), the Attune® cytometer offers a no-wash, no-lyse protocol that avoids cell loss and simplifies sample preparation (Figure 2). This feature is particularly useful for samples that are inherently dilute, such as cerebrospinal fluid (CSF) and stem cells.
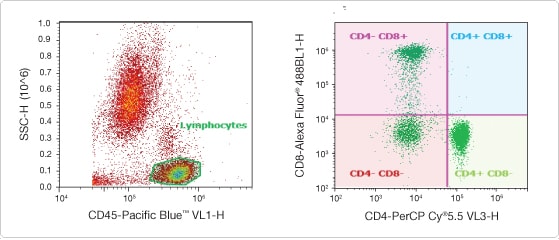
Figure 2. Minimizing sample preparation without compromising data. Normal whole blood (100 μL) was labeled with CD45 Pacific Blue™, CD8 Alexa Fluor® 488, and CD4 PerCP-Cy®5.5 (BD Biosciences) direct conjugates. After 15 min, 5 μL of the stained whole blood was diluted into 4 mL PBS, and data were acquired on the Attune® cytometer without RBC lysis or washing. A fluorescence threshold was set on the Pacific Blue™ dye to include only CD45-positive cells. A gate was created around the lymphocyte population in a CD45 vs. SSC plot (left), to analyze the mutually exclusive CD4 and CD8 populations (right). By diluting the sample and using a rate of 500 μL/min, all sample preparation steps can be eliminated without compromising the data.
Simplified Sample Preparation Delivers Additional Time Savings
With the Attune® cytometer, it is now practical to process stained whole blood without the need to either lyse red blood cells (RBCs) or wash to remove RBC fragments and platelets. In Figure 2, whole blood was simply stained with directly conjugated antibodies, diluted 1:400 in phosphate-buffered saline (PBS), and analyzed on the Attune® cytometer by using a fluorescence threshold on CD45. A cell-permeant nucleic acid dye from our Vybrant® DyeCycle™ stain product line can alternatively be used to select nucleated cells from the core stream by using a fluorescence threshold (Figure 3).
By eliminating lysis and wash steps, this no-lyse, no-wash method for the Attune® cytometer offers time savings and avoids sample loss that can affect cell subpopulations of interest.
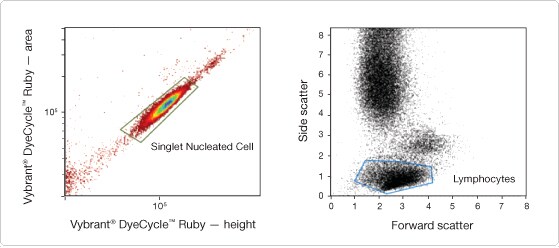
Figure 3. Density plot of Vybrant® DyeCycle™ Ruby–stained human peripheral blood. (Left) Fluorescence height vs. area with a polygon gate selecting single nucleated cells. (Right) A daughter dot plot of the singlet nucleated cell gate is used to gate on the subpopulation of interest. In this case, a polygon gate is set around the lymphocyte population.
References
- Gratama JW, Menéndez P, Kraan J et al. (2000) Loss of CD34(+) hematopoietic progenitor cells due to washing can be reduced by the use of fixative-free erythrocyte lysing reagents. J Immunol Methods 239:13–23.
- Weaver JL, McKinnon K, Germolec DR (2010) Phenotypic analysis using very small volumes of blood. Curr Protoc Cytom 6.30.1–6.30.8.
- Hoff J (2000) Methods of blood collection in the mouse. Lab Animal 29:47–53.
- Waynforth HB, Flecknell PA (1992) Experimental and Surgical Technique in the Rat, Second edition. Academic Press Ltd.
- Roederer M (2002) Compensation in flow cytometry. Curr Protoc Cytom 22:1.14.1–1.14.
Get a copy of this article as it appears in the print version of BioProbes 66.
For Research Use Only. Not for use in diagnostic procedures.