Search
Your DNA may not be accurately quantitated
DNA degradation can result from:
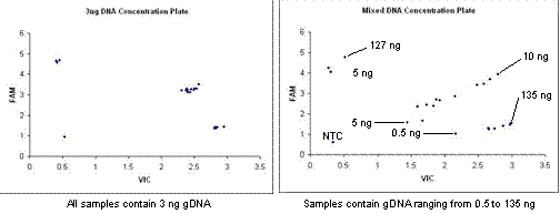
The amount of genomic DNA is critical to the success of the assays. Within an assay and/or study, uniformity of genomic DNA concentration leads to accurate, robust, and reproducible results, and ensures efficient use of valuable samples. Variability in genomic DNA concentrations can lead to experimental anomalies that may affect interpretation of genotyping results, as shown in the following figure. Precise handling and quantitative measurements before running an assay can prevent possible errors without waste of reagents and samples.
Allelic discrimination plots for the TaqMan® Genotyping Assay C_1204092_20
Solution:
- Always perform concentration measurements before using any genomic DNA, even commercially prepared DNA.
- Use the recommended amount of genomic DNA (3 to 20 ng) per sample per assay.
- Always use the same quantity of genomic DNA for all samples of an assay on a plate.
Commercially purchased DNA comes with concentration information, but it is good practice to confirm DNA concentrations in your laboratory. Applied Biosystems scientists have found that the concentrations of DNA listed for commercially available genomic DNA can be quite different from laboratory measurements.
There are numerous methods for quantitating genomic DNA, including:
- UV spectroscopy
- Absolute quantitation using a known copy number gene such as TaqMan® RNase P
- Fluorometric analysis
Applied Biosystems recommends UV spectroscopy or the TaqMan® RNase P method for DNA quantitation.
UV Spectroscopy
UV Spectroscopy is the most widely used method for quantifying DNA of all types. Even though it is most widely used, the consumable reagents used in the process vary greatly.
- Ensure that the spectrophotometer is set up correctly for the reagents to be used.
- Background constants for optical plastic cuvettes and plates are different from those of quartz cuvettes and plates—consult the instrument manual for ways to determine background constant.
- Be cautious of the diluents used with the genomic DNA samples. They can have differing properties which may affect the final results.
- Use UV spectroscopy can to quantitate genomic DNA by reading sample absorbance at 260 nm (A260). The A260 is most accurate when your nucleic acid is pure, and is most useful for DNA in microgram quantities (Gallagher 1994).
- Take a concurrent reading at 280 nm to determine the concentration of contaminating proteins—the A260/A280 ratio is used to determine purity of a DNA sample. Proteins, particles in the solution, and aromatic chemicals can affect the reading.
The effective read range of UV spectroscopy is 0.1 to 0.999 which corresponds to approximately 4 ng/µL to 50 ng/µL of genomic DNA. Values above or below that range are invalid absorbance readings.
- To ensure accurate quantitative results, dilute your genomic DNA samples so that the A260 reading is between 0.1 and 0.999 (remember to record the dilution factor and the diluents used).
- Ensure that there is enough genomic DNA to use this method—most plates and cuvettes have minimum working volumes, and the genomic DNA sample used for the quantitative measurement will be discarded.
Absolute Quantitation
Absolute quantitation measures the total amount of amplifiable genomic DNA. This technique requires the creation of a standard curve using genomic DNA samples of known concentrations. The standard samples must be pre-quantitated and validated using an independent method such as spectrophotometry or fluorometry. The unknown samples are compared to the known samples for quantitation.
Two well-known techniques for absolute quantitation are:
- TaqMan® Assay Chemistry
- SYBR® Green Assay
Absolute quantitation using the TaqMan® technology is a highly accurate technique for quantifying DNA. The TaqMan® DNA Template Reagents and the TaqMan® RNase P Detection Reagents provide convenient methods to quantitate genomic DNA. The kits include pre-diluted and validated standards at five concentrations per kit: 0.6 ng/µL, 1.2 ng/µL, 3.0 ng/µL, 6.0 ng/µL, 12.0 ng/µL. Dilute or aliquot to the appropriate range for the samples.
SYBR® Green is a dye that is a minor groove binder which binds only to double-stranded DNA (dsDNA). This method is less specific than the TaqMan® method because the dye will bind to any dsDNA. The use of this method will require melt curve analysis to verify the specificity of the assay.
For either technique, be sure to run the standard curve and unknown samples on the same plates in the SDS instrument.
Fluorometric Analysis
Quantitation of DNA by fluorometric analysis uses various intercalating dyes. These are summarized in the table below:
Dye features
| Dye | Features |
|---|---|
| Hoechst dye #33258 | More sensitive than spectrophotometric measurements due to low affinity for RNA * Base composition of the DNA can affect readings because the dye binds preferentially to AT-rich DNA (Gallagher, 1994) |
| Ethidium bromide | Not base composition sensitive Binds to RNA Capable of detecting nanogram amounts of DNA Ideal if the DNA is relatively pure with a high GC content (Gallagher, 1994) |
| Pico Green | Can quantitate as little as 25 pg/mL and up to 100 ng/mL of dsDNA |
For Research Use Only. Not for use in diagnostic procedures.