Search
How a Flow Cytometer Works
Cytometry, in its purest form, is the measurement of cell characteristics, which can include cell size, cell count, cell cycle and more. This technique allows researchers to get highly specific information about individual cells. Typical samples include everything from a cell line expressing Green Fluorescent Protein to heterogeneous cell populations harvested from tissues. The most critical requirement for efficient and effective flow cytometry analysis is that the sample be a single-cell suspension. This helps ensure that every cell is analyzed independently.
Topics in this section:
Introduction to flow cytometry
Flow cytometry is a cell analysis technique that was first used in the 1950s to measure the volume of cells in a rapidly flowing fluid stream as they passed in front of a viewing aperture. Since that time, innovations from many engineers and researchers have culminated in the modern flow cytometer, which is able to make measurements of cells in solution as they pass by the instrument’s laser at rates of 10,000 cells per second (or more). Today’s instruments offer an increased number of detectable fluorescent parameters (from 1 or 2 up to ~30 or more), all measured at the same time on the same cell. Because of its speed and ability to scrutinize at the single-cell level, flow cytometry offers the cell biologist the statistical power to rapidly analyze and characterize millions of cells, albeit at the expense of the morphological characteristics and subcellular localization that microscopy can provide. Science, like the rest of life, is all about trade-offs! See Table 1 for a comparison of flow cytometry and microscopy techniques.
Table 1. Comparing flow cytometry and fluorescence imaging microscopy as techniques for cellular analysis.
| Flow cytometry | Microscopy | Comments | |
|---|---|---|---|
| General | |||
| Instrumentation/software complexity and expense | +++ | +++ | Flow cytometry: the more parameters needed the more complex and expensive the instrumentation/analysis software will be Microscopy: relatively inexpensive but again, the more complex the experiment the more likely the instrument and analysis software will be more expensive |
| Number of parameters detected on single cell | +++ | + | Flow cytometry: up to 30 parameters Microscopy: up to 6 parameters with special instruments |
| Quantitation | +++ | ++ | Flow cytometry: easily obtain statistics using embedded software Microscopy: can be achieved with instrumentation and software, or can sometimes be done manually but is time consuming |
| Sensitivity | +++ | +++ | Flow cytometry: dependent on the fluorochromes, experimental design, and instrumentation Microscopy: dependent on fluorochrome choice and exposure time |
| Throughput | +++ | ++ | Flow cytometry: millions of cells in short time Microscopy: requires the use of specific imaging instrumentation for high throughput |
| Sample types | |||
| Fixed cells | +++* | +++ | Can be analyzed with either technique |
| Live cells | +++ | +++ | Can be analyzed with either technique |
| Tissue | + | +++ | Flow cytometry: requires tissue dissociation Microscopy: may require tissue sectioning but in situ characteristics can be detected |
| Applications | |||
| Cell sorting | +++ | - | Flow cytometry (via specialized flow cytometer called a cell sorter): physical separation and collection of user-defined cell populations Microscopy: not possible |
| Kinetic/time-lapse data | + | ++ | Flow cytometry: possible but challenging Microscopy: routinely done |
| Rare cell detection | +++ | + | Flow cytometry: easily performed Microscopy: time consuming to identify without software or instrumentation |
| RNA | ++ | ++ | Flow cytometry: possible with specialized assays that combine protein and RNA detection together in the same assay Microscopy: easily visualized, new assays allowed to develop multiparametric analysis |
| Structural/morphological data | + | +++ | Flow cytometry: imaging cytometers are now available that combine flow cytometry with imaging Microscopy: major strength of this technique |
| * Assumes reagents have been validated to work for fixed cells. | |||
Potential applications for flow cytometry
The potential applications of analysis by flow cytometry are numerous, including the detection and measurement of:
- Protein expression—throughout the entire cell, even the nucleus
- Protein post translational modifications—includes cleaved and phosphorylated proteins
- RNA—including IncRNA, miRNA, and mRNA transcripts
- Cell health status—from viability to late-stage apoptosis or programmed cell death
- Cell cycle status—providing a powerful tool to assess cells in G0/G1 phase versus S phase, G2, or polyploidy, including analysis of cell proliferation and activation
- Identification and characterization of distinct subsets of cells within a heterogeneous sample—including distinguishing central effector memory cells from exhausted T cells or even regulatory T cells
An additional capability of specialized flow cytometers is the ability to sort cells and recover the subsets for post experimental use. This specialized flow cytometer is called a fluorescence activated cell sorter (FACS), a term that is sometimes erroneously used interchangeably with ‘flow cytometer’. This usage is incorrect. A flow cytometer is an analytical machine that can perform cell sorting when configured with the appropriate hardware and software as found in FACS. Cell sorters use fluidics and fluorescence components similar to those in flow cytometers, but are able to divert a specific population from within a heterogeneous sample into a separate tube, typically based on specified fluorescence characteristics. If collected under sterile conditions, these cells can be further cultured, manipulated, and studied.
Overview of how a flow cytometer works
The three main components of a flow cytometer are the fluidics, optics, and electronics (Figure 1).
- The fluidics system of a flow cytometer is responsible for transporting sample from the sample tube to the flow cell. Once through the flow cell (and past the laser), the sample is either sorted (in the case of cell sorters) or transported to waste.
- The components of the optical system include excitation light sources, lenses, and filters used to collect and move light around the instrument and the detection system that generates the photocurrent.
- The electronics are the brains of the flow cytometer. Here, the photocurrent from the detector is digitized and processed to be saved for subsequent analysis.

Figure 1. Schematic of the three main components of a flow cytometer.
Sample uptake into the instrument
A typical experiment begins with fluorescently labeled cells in a single-cell suspension, but a sample containing particles of any kind can be used. Once the sample is placed on the flow cytometer, the sample is taken up into the instrument, and the cells are surrounded by a physiological buffer called sheath fluid. The fluidics system—the tubing, pumps, and valves—organizes the initial sample suspension into a single-file stream of cells (Figure 2) as they make their journey through the flow cytometer for analysis.

Figure 2. The fluidics of the flow cytometer. The flow cell (also called the flow chamber) allows the cells (particles) within the samples to line up single file. This is a critical step for single-cell analysis.
Interrogation point
The place where the cells interact with laser light is called the interrogation point (Figure 3). You might also hear it referred to as the laser intercept. This is where the action takes place. When the laser light beam illuminates a single cell, some of the light will strike physical structures within the cell, causing the light to scatter. This light scatter can be measured and correlated with relative cell size and structures inside the cell. These measurements are termed forward angle scatter(FSC) and side angle scatter (SSC), depending on where the light is collected with respect to the path of the laser. Nearly simultaneously, light from the laser will excite all fluorophores associated with the cell, which produces a fluorescence emission. All of this light is collected by the detector and processed through the electronics component of the flow cytometer. After passing through the interrogation point, the cell is no longer needed and is carried by the fluidics system to the waste container. If this were a cell sorter, this would be the point where the cell would be passed to a collection tube and used for further experiments.

Figure 3. The interrogation point. This is the place within the flow cytometer where the laser light hits the individual particles as they pass in front of the laser, one at a time.
Summary
Flow cytometry is a powerful tool that can be used in a significant number of cell analysis applications ranging from phenotyping to cell health and viability.
The two greatest advantages of flow cytometry are its ability to measure a large number of parameters (2 to 30 or more) on the same sample and its ability to collect information from millions of cells in a matter of seconds.
There are 3 main components of a flow cytometer—the fluidics, optics and electronics—that work together to provide a complete system of cell analysis (Figure 4).
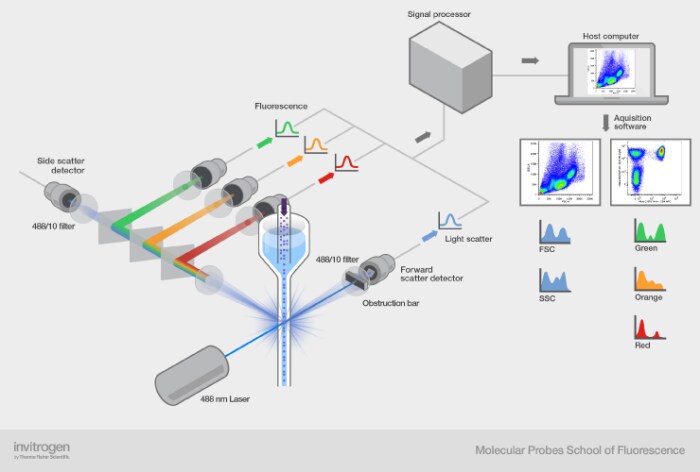
Figure 4. The working parts of the flow cytometer.
For Research Use Only. Not for use in diagnostic procedures.
