Search
Getting Started with Thermo Scientific 1-Step IVT Systems
During development, our R&D scientists had a 95% success rate when expressing 100 different proteins using the Thermo Scientific Human In Vitro Translation System. This overview highlights the critical steps to ensure superior results and expression levels with your unique in vitro translation experiments using either DNA (expression vector or PCR amplicon) or mRNA (in vitro transcription reactions) templates.
View products
Preparing for human in vitro expression
Preparing your gene for in vitro translation begins with identifying the proper elements for successful protein production. While researchers can source a gene from an open reading frame collection, PCR amplification from cells or plasmid, these coding sequences are generally not ready for optimal human in vitro expression. The following section covers the critical elements that must flank a coding sequence to ensure optimal levels of mRNA transcript stability and translation efficiency. These elements can be introduced by cloning a gene of interest into our recommended expression vectors (included in all Pierce Human In Vitro Protein Expression Kits) or by adding the necessary elements through PCR.

Transcription elements
The Human In Vitro Translation System was optimized using cDNAs cloned into the Thermo Scientific pT7CFE1 expression vectors. Below are explanations of the critical elements (especially the IRES element) within this vector that enable efficient in vitrotranslation in our human cell-free system.
RNA polymerase promoter
Bacteriophage RNA polymerases are better suited for fast, repetitive transcription than multi-subunit RNA polymerases from bacteria or higher organisms. Monomeric bacteriophage RNA polymerases are ideal for in vitro transcription reactions because of their high efficiency and relatively simple structure. For this reason, a T7 bacteriophage RNA polymerase was chosen for transcription in the Human IVT System. Therefore, in vitro transcription reactions using the Human In Vitro Expression System require the T7 RNA polymerase promoter element to be inserted upstream of the coding sequence in order to generate mRNA. The pT7CFE1-CHis Expression Vector (included with Human IVT Kits) possesses a T7 RNA polymerase promoter that functions with the T7 RNA polymerase included in the transcription reaction mixture.
Note: In pT7CFE1-CHis, the T7 RNA polymerase promoter sequence (nucleotides 1-18) is 5'-taatacgactcactatag-3'
Transcription terminator
Sequences containing transcription terminators mark the end of the gene for transcription machinery. This element is most important when very long non-coding sequences follow the end of the mRNA coding region or circular plasmid templates. In addition to producing mRNAs with more uniform length and stability, the terminator sequence helps preserve the transcription reaction materials (NTPs) for the synthesis of coding sequences.
Note: In pT7CFE1-CHis, the T7 terminator sequence (nucleotides 700-747) is 5'-tagcataaccccttggggcctctaaacgggtcttgaggggttttttg-3'
Translation elements for high yield
IRES (high yield ribosomal binding sequence)
All eukaryotic mRNAs in vivo have a 5' cap structure that contains a 7-methylguanosine (m7G) linked to the first nucleotide of the mRNA through a 5'-5' triphosphate bridge. The 5' cap promotes the efficient translation and increased half-life of mRNAs by preventing the exonucleolytic degradation of the transcript.
Alternatively, the internal ribosome entry site (IRES) from the Encephalomyocarditis virus (EMCV) can be substituted for a 5' cap. The unique secondary structure of the IRES element enables very efficient recruitment of ribosomes to mRNA and translation initiation. This property enables very high levels of protein expression from mRNA containing the IRES element in the absence of a 5' cap. All T7 RNA polymerase mRNA transcribed using the pT7CFE1-CHis expression vector will possess an IRES element to promote optimal expression levels when used with the Human IVT System.
Important note regarding the IRES element: The 1-Step Human IVT Kit can be used with DNA or RNA templates. If you already have mRNA generated from an alternative expression vector that does not contain an IRES element, protein yield will be greatly reduced. You can increase the stability and translation efficiency by capping the mRNA. Kits are commercially available for capping mRNAs in vitro, but the expression levels of 5' capped mRNA will still be greatly reduced compared to using the IRES element. In the absence of either an IRES element or a 5' cap, in vitro translation will likely fail (see data).
Note: In pT7CFE1-CHis, the EMCV IRES sequence includes nucleotides 33 to 535

View products
Kozak sequence
The Kozak consensus sequence [(gcc)gccRccAUGG] found in eukaryotic mRNAs plays a major role in the translation initiation process. In mRNA transcribed from pT7CFE1-CHis, the IRES element functions as the ribosomal binding site, whereas the Kozak sequence helps orient the ribosome to the correct ATG codon for translation initiation. A prokaryotic Shine-Dalgarno sequence will not work in eukaryotic in vitrotranslation systems.
Note: The Kozak sequence (nucleotides 524-536) in pT7CFE1-CHis is 5'-gatgatAatATGG-3'
Fusion tags
Affinity tags can greatly simplify applications that require the purification of recombinant protein. To aid in these efforts, pT7CFE1-CHis contains a C-terminal histidine tag to facilitate the purification and detection of full-length recombinant proteins. For protein expression without the histidine tag, ensure that a stop codon is located immediately after the last amino acid codon of your cDNA.
Note: In pT7CFE1-CHis, the 6xHis coding sequence (nucleotides 611-628) is 5'-caccaccaccaccaccac-3'
Learn more
Polyadenosine tail
The polyadenosine tail (poly(A) tail) at the 3'-end of eukaryotic mRNAs is a stretch of up to 200-300 adenosine ribonucleotides added downstream of the translation termination codon. (Exception: 3' ends of histone mRNAs have a stem and loop structure plus a purine-rich sequence instead of a poly(A) tail). Polyadenylation is known to protect mRNA from enzymatic degradation and also aids in translation. A stretch of 30 adenosine ribonucleotides at the 3' end of mRNA is sufficient to stabilize transcripts for in vitro translation.
Note: In pT7CFE1-CHis, the poly(A) tail sequence (nucleotides 642-671) is 5'-aaaaaaaaaaaaaaaaaaaaaaaaaaaaaa-3'
Generating mRNA

Cloning into the pT7CFE1-CHis expression vector
The pT7CFE1-CHis expression vector is optimized for expression in the Human In VitroTranslation System because it possess all of the proper elements. The multiple cloning site has 11 unique restriction enzyme sites to allow the selection of sites that are not found in your cDNA sequence. To ensure proper translation, make certain that your cloned cDNA is in frame with the "ATG" start codon that lies upstream of the Msc1 restriction enzyme site. The pT7CFE-CHis vector also contains a C-terminal 6X histidine tag to assist in purification. If a C-terminal histidine tag is not desired in the final product, an "AUG" stop codon should be located after the coding sequence of your cDNA in order to properly terminate translation.
Note: In pT7CFE1-CHis, the multiple cloning site (nucleotides 550-610) sequence is5'-tggccaccacccatatgggatccgaattcgatatcttaatt
aagctgcaggagctcgtcgacgcggccgcactcgag-3'
Note: In pT7CFE1-CHis, the stop codon after the 6xHis coding sequence (nucleotides 629-631) is 5'-tga-3'

View products
Addition of the proper transcription and translation elements by PCR
When not using pT7CFE-CHis or an expression vector with the same required elements, in vitro protein expression is still possible by adding the proper transcription and translational elements to your cDNA by PCR (add link to PCR tech tip). This two-step PCR protocol outlined below allows researchers to skip cloning prior to protein expression and achieve high protein yield. This method is effective for using any plasmid or open reading frame as the starting template.
The upstream elements added to a gene by this PCR method include a T7 promoter, IRES element and Kozak sequence. To improve mRNA stability, a poly(A) sequence (21nt) also can be added to the 3' end of the cDNA. The PCR product can either be purified or directly used for transcription. The mRNA generated from the transcription can then be added to a translation reaction mix containing all of the machinery for protein expression.
A sample protocol for performing these steps described above can be found in a downloadable tech tip (Tech Tip #72).
Learn more
Setting up positive controls
Each Human Coupled IVT Kit comes complete with a positive control vector to help you monitor the success of your procedure. The included controls are either an expression vector for Turbo Green Fluorescent Protein (tGFP) or a tGFP mRNA (included in the DNA or RNA expression kits, respectively). For both types of expression kits, a GFP expression reaction is performed in parallel with your desired protein sample. Successful expression of the GFP positive control may be assessed qualitatively (visual detection) or quantitatively (spectroscopic detection).
Visual detection
A 25µL GFP translation reaction can be visualized directly in the reaction microcentrifuge tube. Because components of the Human In Vitro Translation System do not quench fluorescent signal, clean-up steps are not necessary prior to detecting fluorescence. This is not possible with rabbit reticulocyte lysates, which do quench fluorescent signal. GFP fluorescence detection can be performed using a microscope or gel imager equipped with a FITC filter (excitation/emission: 482nm/512nm).

Spectroscopic detection
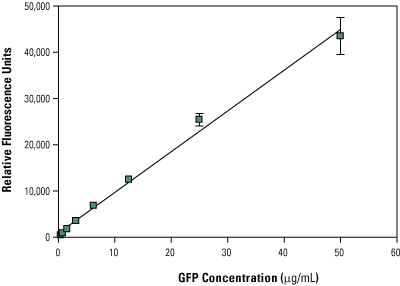
To calculate GFP positive control protein expression yield, create a standard curve using recombinant GFP (Part No. 88899). To do this, set up serial dilutions of recombinant GFP in PBS ranging from 0.40µg/mL to 50µg/mL. Samples can be read in either 96- or 384-well formats on a fluorescent plate reader equipped with a FITC filter (excitation/emission: 482nm/512nm). Measure the fluorescent signal of the GFP standards (known concentration) and the GFP translation reaction (unknown concentration) to determine protein expression yield (µg/mL). Plot the results of the standard curve and extrapolate results of the unknown GFP translation control. Typical GFP expression levels for a 90-minute translation reaction are approximately 25µg/mL.
View products
For Research Use Only. Not for use in diagnostic procedures.