Search
Automation of One-Step RT-PCR Using Ambion's Cells-to-cDNA™ II Kit and the MultiPROBE® II HT Liquid Handling System
Additional Authors: Brittan Pasloske Ph.D., Nathan Harris*, Jennifer Van Dinther*
*PerkinElmer Life Sciences, 2200 Warrenville Road, Downers Grove, IL 60515
Overview
- Automated protocol for one-step RT-PCR on tissue cultured cells, eliminating need for a separate RNA isolation step
- Cells are washed, lysed, DNase treated and prepared for one-step RT-PCR using the MultiPROBE Liquid handling system
- Sensitive and reproducible measurement of gene expression in cultured cells
- Compatible with real-time RT-PCR
Introduction
High throughput RT-PCR analysis of RNA from cells grown in tissue culture is an expensive and multi-step process mainly because of the required up-front RNA isolation. As well, RNA isolation is generally the rate-limiting step in such experiments.
Cells-to-cDNA™ II technology (US patent pending) is designed to completely bypass RNA isolation thereby increasing the throughput potential and decreasing overall costs. With a novel cell lysis buffer, cells are lysed and RNases are inactivated in a single step yielding a cell lysate that is competent for reverse transcription.
Ambion, Inc. and PerkinElmer Life Sciences now offer an automated protocol for the Cells-to-cDNA II technology using the MultiPROBE® II HT Liquid Handling System and Gripper Integration Platform (PerkinElmer Life Sciences). This automated protocol includes cell lysis, DNase I treatment, and set-up for one-step RT-PCR, that can then be used in gel-based or quantitative (real-time) PCR assays all in a 96-well format.
Data is presented showing the quantitative analysis of RNA from cultured cells in a 96-well format demonstrating reproducibility and consistency of results using the MultiPROBE® II HT Liquid Handling System. An experiment showing the stimulation of the plasminogen activator gene (t-PA) by exposure to phorbol myristate acetate (PMA) provides an example of how this technology can be used.
Experimental - Equipment and Materials
MultiPROBE II HT EX
8-probe Varispan Pipetting 
Figure 1. MultiPROBE II HT EX Running Cells-to-cDNA™II Protocol.
Gripper™ Integration Platform
- Gantry-based system with 5 axes of motion
Cells-to-cDNA™II
- Complete reverse transcription kit including 1X PBS, Cell Lysis Buffer, DNase I, and both an Armored RNA® and endogenous controls
- Cell lysates are used directly in reverse transcription eliminating the RNA isolation step
- Easily adaptable to a one-step RT-PCR procedure
Method
Cell Preparation
- HeLa S3 cells are grown overnight in 0.2 ml DME media with 10% FBS with equal number of cells in each well.
- After an overnight incubation, phorbol myristate acetate (PMA) is added to final concentrations of 100 nM, 10 nM, 1 nM, 0.1 nM, and 0 nM in the growth media in replicates of eight. Incubate at 37°C for 24 hr.
Automated Protocol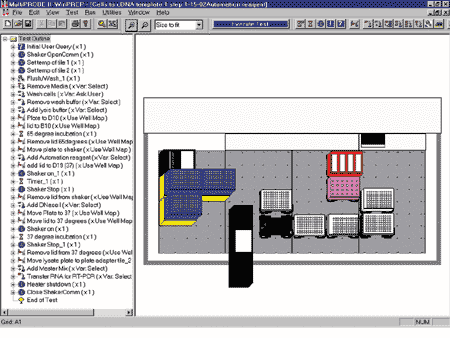
Figure 2. Screen View Using WinPREP™ Software. Screen Left: outline of protocol using Cells-to-cDNA II. Screen Right: view of deck layout. - Place 96-well plate and lid on the MultiPROBE® II HT Liquid Handling System on the proper deck positions.
- Remove the growth media and wash the cells with 1X PBS.
- Add 0.1 ml cell lysis buffer to each well.
- Move plate to 75°C heating tile and incubate for 10 min.
- Move plate to shaker platform and add 2 µl Lysis Buffer II to each well. Shake for 2 min.
- Add 2 µl DNase I to each well and move plate to 37°C heating tile. Incubate for 15 min while shaking.
- Move plate to 75°C heating tile and incubate for 5 min to inactivate the DNase I.
- Prepare a master mix for one-step RT-PCR.
- Aliquot 20 µl of the master mix to a 96-well PCR plate.
- Add 5 µl of lysate to each well. The plate is now ready for one-step RT-PCR. PCR
- Setup a PCR profile for an ABI 7700 as follows:
42°C for 15 minutes (reverse transcription)
94°C for 3 minutes (denaturation)
40 Cycles: 94°C for 20 seconds
60°C for 40 seconds
Results and Analysis
Figure 3 shows a run with Cells-to-cDNA II using the automated protocol on 88 samples from a 96-well culture plate to detect GAPDH. 
Figure 3. Real-time Quantitation of GAPDH Using the Automated Profile.
Mean = 17.28
Standard Deviation = 0.524
CV = 3.03%
N = 88
High = 18.88
Low = 15.90
A standard curve for both t-PA and 18S rRNA was used to determine relative amounts of both t-PA and 18S rRNA in each sample. Data shows a 29-fold stimulation of t-PA gene by a 24 hr induction of PMA at 100 nM (Figure 4A) while the level of 18S rRNA stayed the same (Figure 4B). These differences are summarized in the chart in Figure 4C. 
Figure 4A. Real-time Quantitation of t-PA Levels After 24 Hour PMA Induction. 
Figure 4B. Real-time Quantitation of 18S rRNA Levels After 24 Hour PMA Induction. 
Figure 4C. Relative Levels of 18S and t-PA with Different PMA Concentrations.